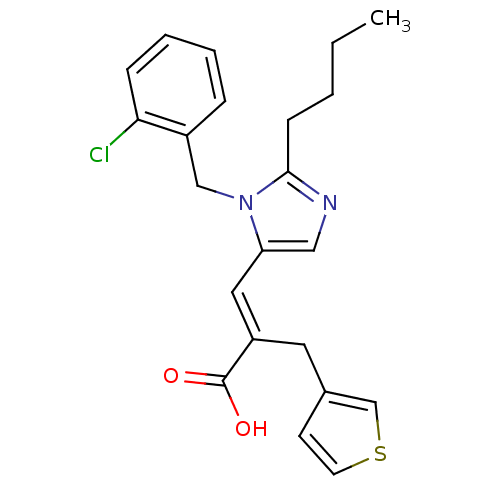
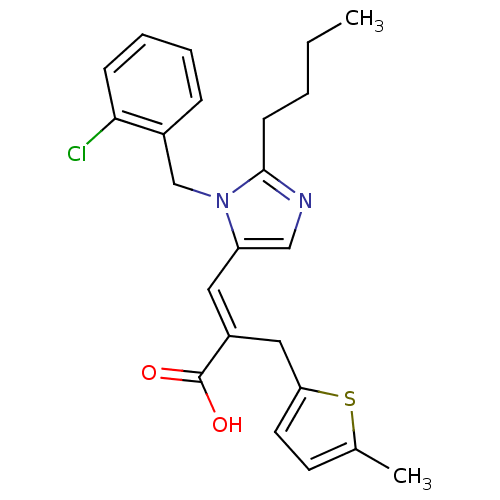
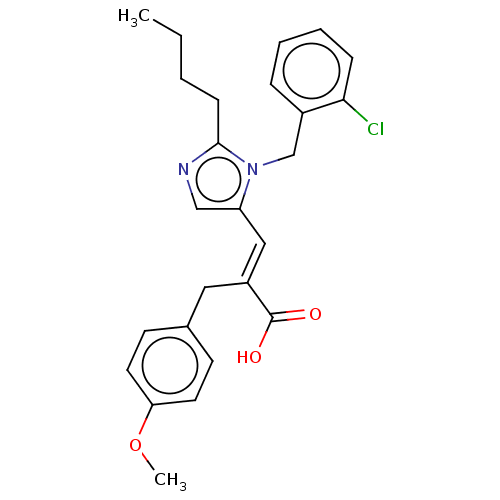
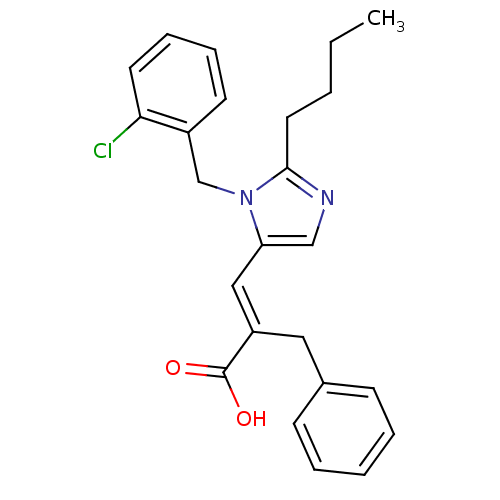
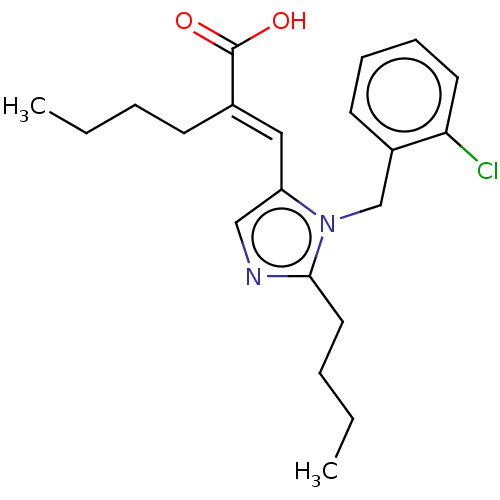
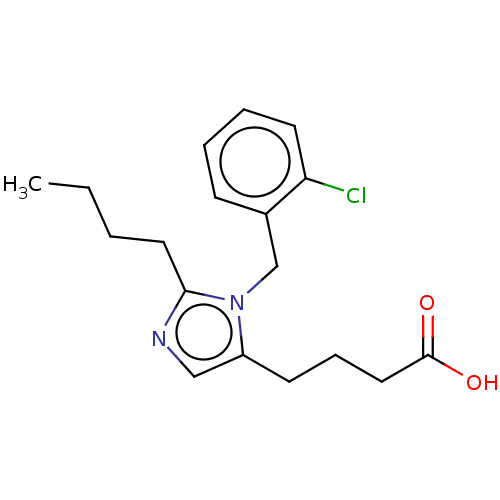
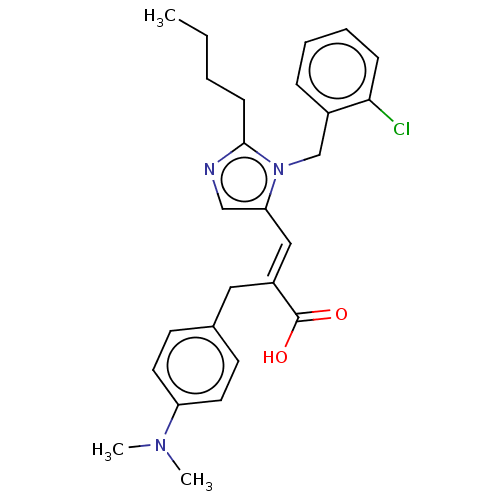
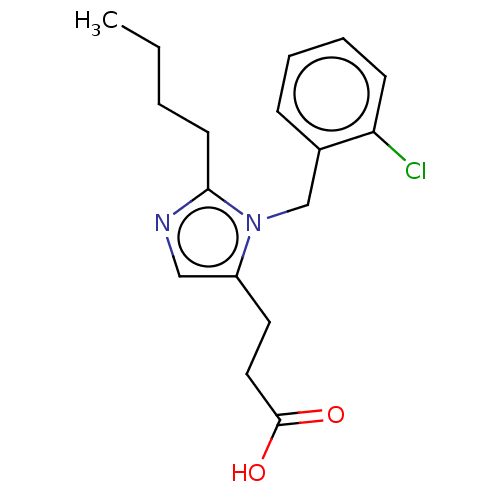
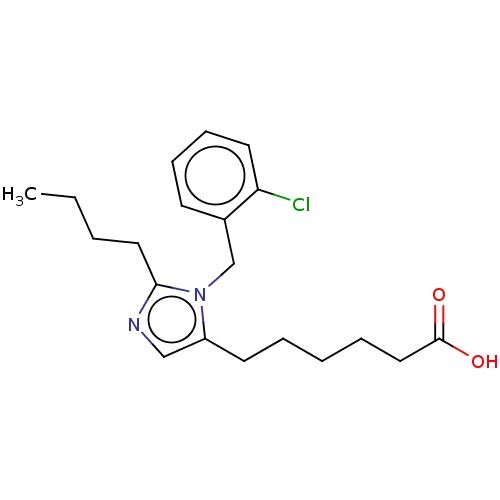
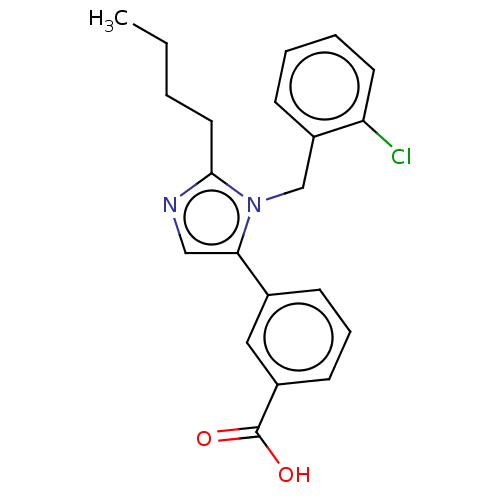
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 58nMAssay Description:Inhibition of human carbonic anhydrase IIMore data for this Ligand-Target Pair
Affinity DataIC50: 90nMAssay Description:Inhibition of [125I]-Angiotensin II receptor specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 120nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 150nMAssay Description:Inhibition of human carbonic anhydrase IIMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 250nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 300nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:Inhibition of [125I]-Angiotensin II receptor specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
Affinity DataIC50: 330nMAssay Description:Inhibition of [125I]-Angiotensin II receptor specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 360nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
Affinity DataIC50: 400nMAssay Description:Inhibition of [125I]-Angiotensin II receptor specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
Affinity DataIC50: 440nMAssay Description:Inhibition of [125I]-Angiotensin II receptor specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 470nMAssay Description:Inhibition of human carbonic anhydrase IIMore data for this Ligand-Target Pair
Affinity DataIC50: 700nMAssay Description:Inhibition of [125I]-Angiotensin II receptor specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
Affinity DataIC50: 730nMAssay Description:Inhibition of [125I]-Angiotensin II receptor specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 810nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 810nMAssay Description:Inhibition of human carbonic anhydrase IIMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 820nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 860nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 1.20E+3nMAssay Description:Inhibition of human carbonic anhydrase IIMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 1.90E+3nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
Affinity DataIC50: 2.40E+3nMAssay Description:Inhibition of [125I]-Angiotensin II receptor specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
Affinity DataIC50: 2.60E+3nMAssay Description:Inhibition of [125I]-Angiotensin II receptor specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 2.90E+3nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of [125I]-Angiotensin II receptor specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 3.60E+3nMAssay Description:Inhibition of human carbonic anhydrase IIMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 4.60E+3nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibition of [125I]-Angiotensin II receptor specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 5.20E+3nMAssay Description:Inhibition of human carbonic anhydrase IIMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 6.80E+3nMAssay Description:Inhibition of human carbonic anhydrase IIMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 6.90E+3nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 8.90E+3nMAssay Description:Inhibition of human carbonic anhydrase IIMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 9.80E+3nMAssay Description:Inhibition of human carbonic anhydrase IIMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Compound was evaluated for the inhibition of [125I]-AII specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+4nMAssay Description:Compound was evaluated for the inhibition of Angiotensin II receptor induced vasoconstriction of the rabbit aortaMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 1.20E+4nMAssay Description:Inhibition of human carbonic anhydrase IIMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 1.46E+4nMAssay Description:Inhibition of human carbonic anhydrase IIMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 1.65E+4nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 1.70E+4nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 1.87E+4nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 1.90E+4nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 2.01E+4nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 2.19E+4nMAssay Description:Inhibition of human carbonic anhydrase IIMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 2.20E+4nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 2.23E+4nMAssay Description:Inhibition of human carbonic anhydrase IIMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 2.35E+4nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
Affinity DataIC50: 2.56E+4nMAssay Description:Inhibition of [125I]-Angiotensin II receptor specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 2.84E+4nMAssay Description:Compound was tested in vitro for binding affinity against human carbonic anhydrase II; (ki*10e-9)More data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 3.39E+4nMAssay Description:Inhibition of [125I]angiotensin II specific binding to rat mesenteric arteriesMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor A/Type-1 angiotensin II receptor B/Type-2 angiotensin II receptor(RAT)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 3.80E+4nMAssay Description:Inhibition of human carbonic anhydrase IIMore data for this Ligand-Target Pair