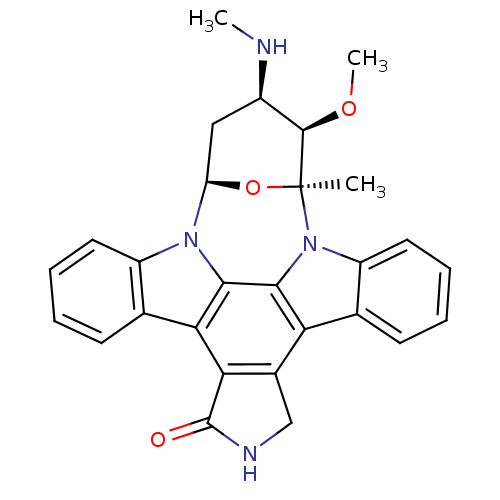
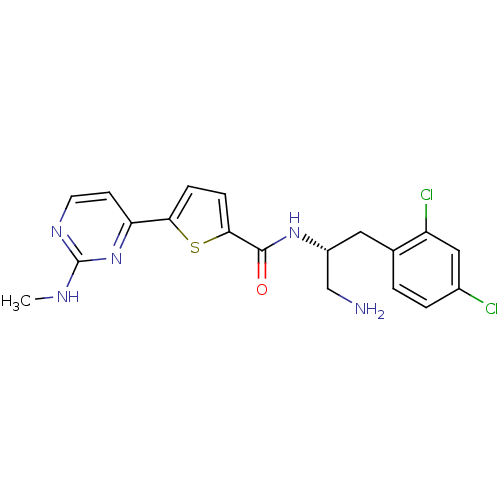
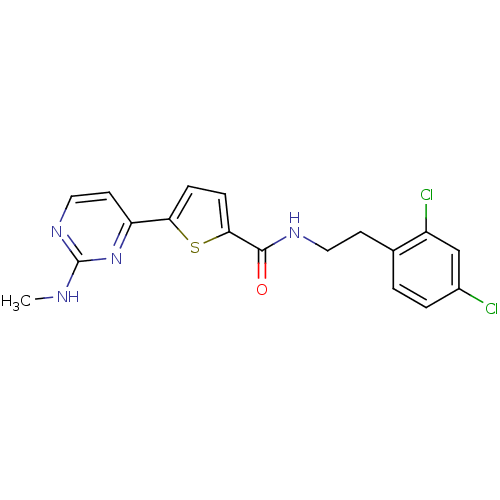
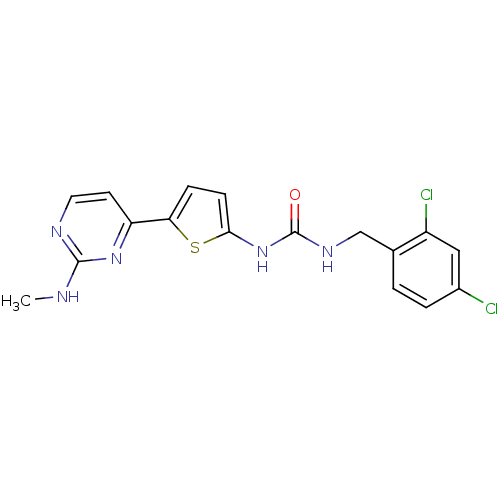
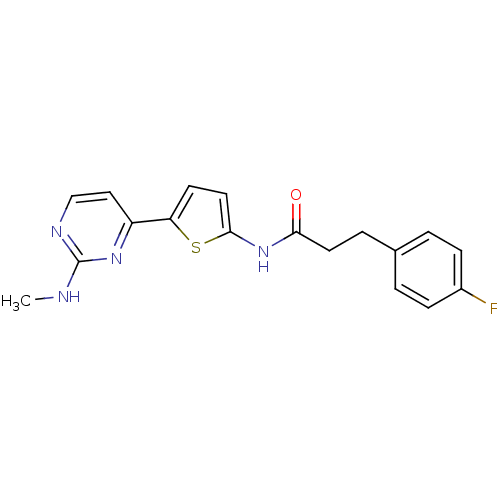
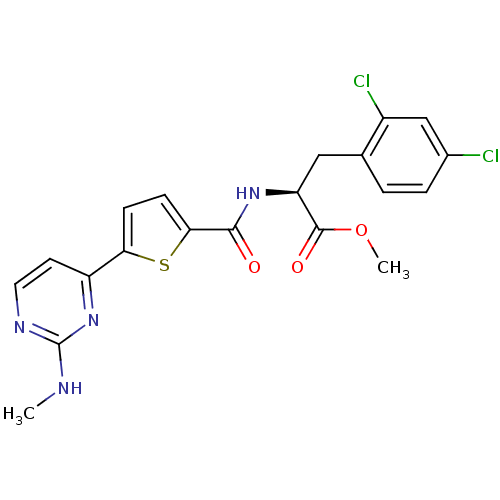
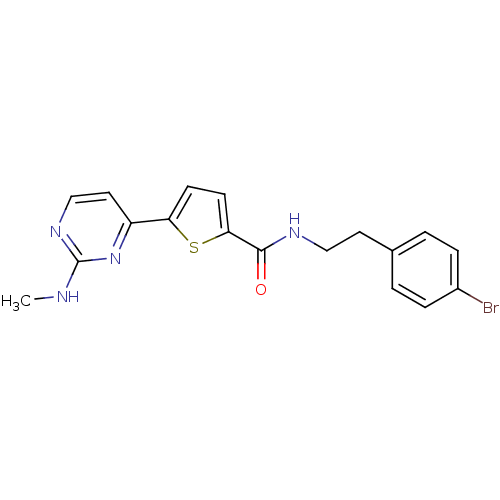
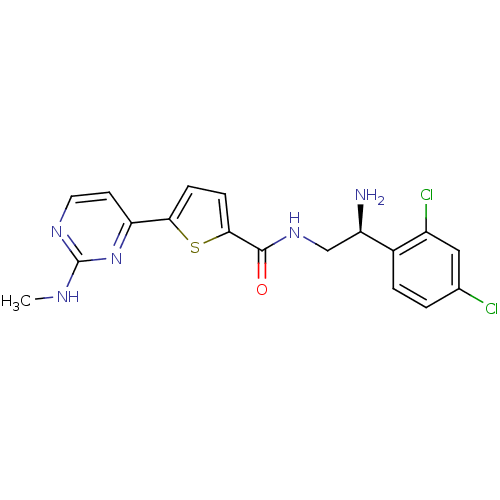
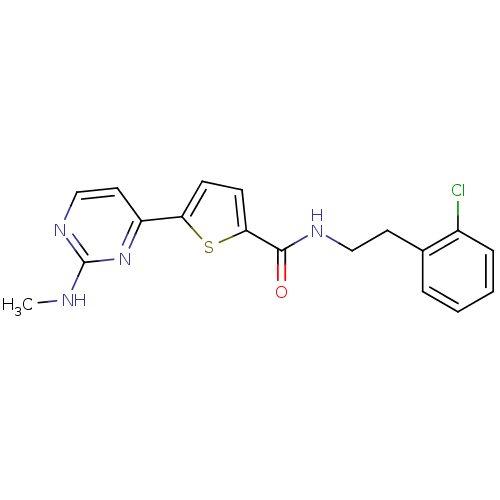
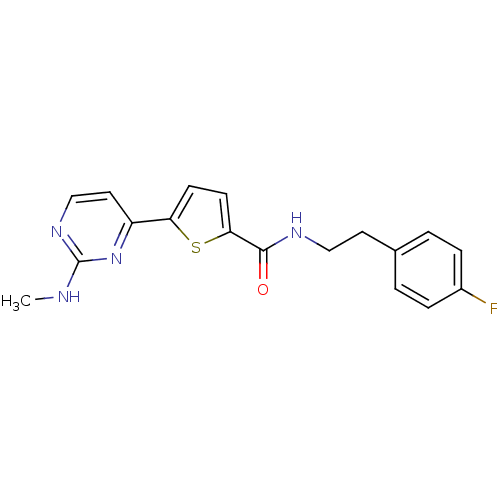
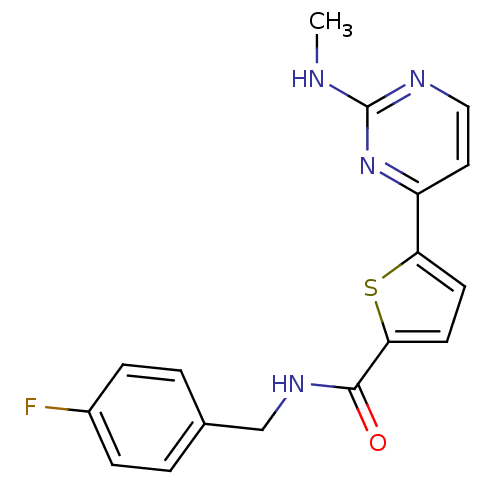
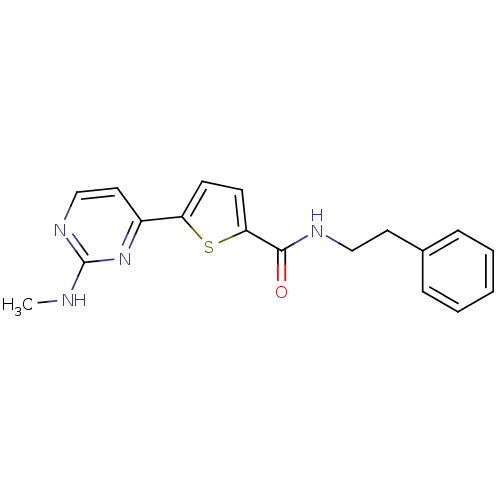
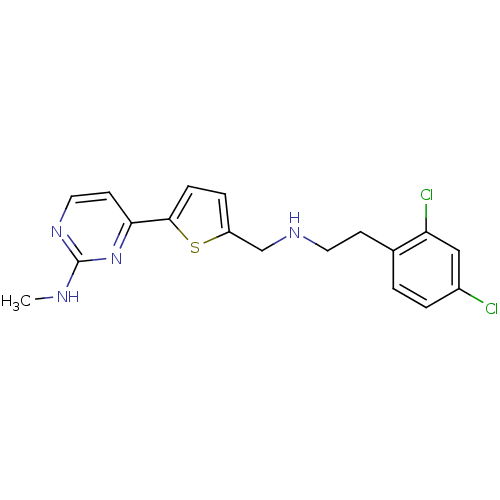
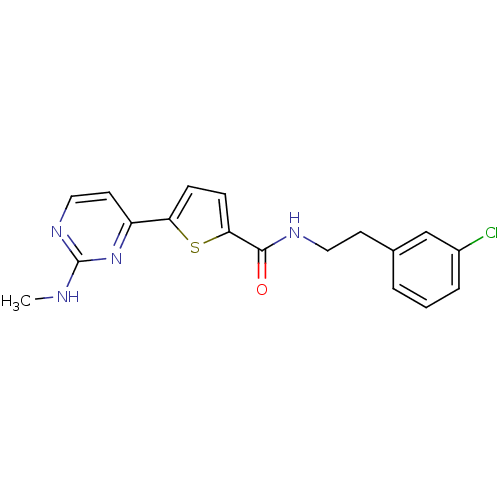
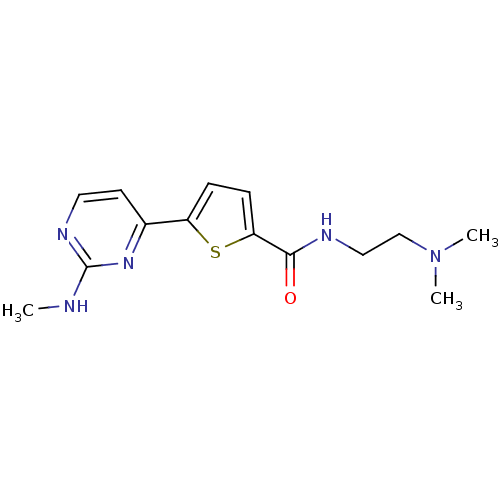
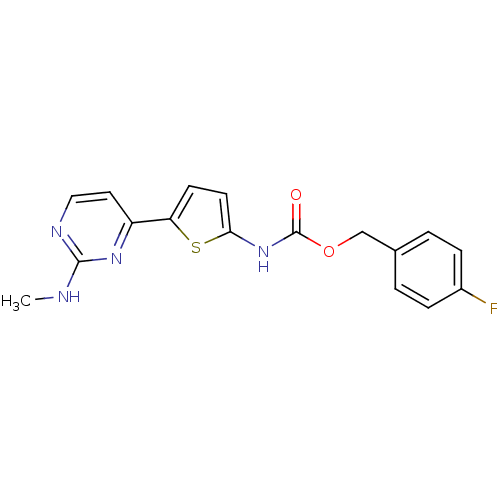
TargetcAMP-dependent protein kinase catalytic subunit alpha/beta/gamma(Homo sapiens (Human))
Chiron
Curated by ChEMBL
Chiron
Curated by ChEMBL
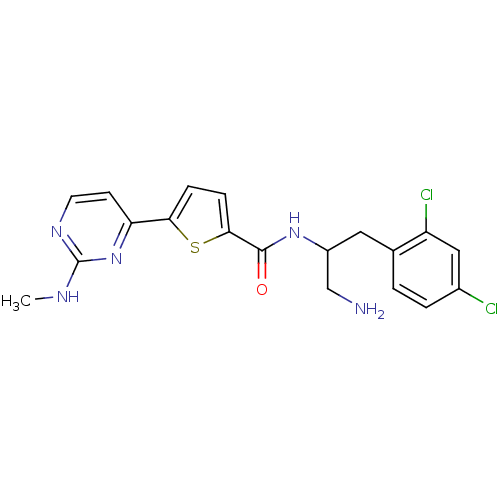
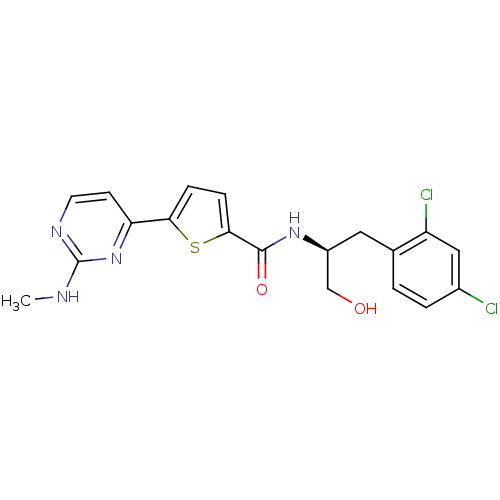
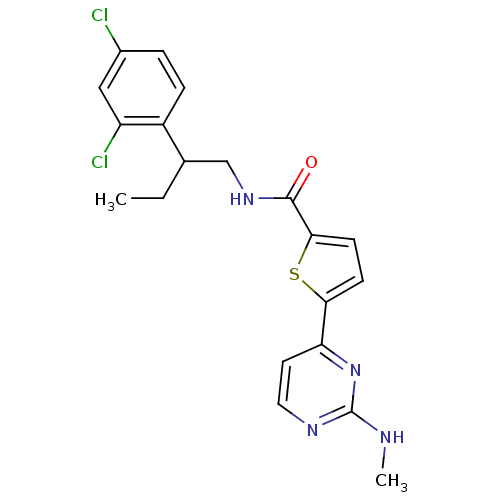
TargetcAMP-dependent protein kinase catalytic subunit alpha/beta/gamma(Homo sapiens (Human))
Chiron
Curated by ChEMBL
Chiron
Curated by ChEMBL
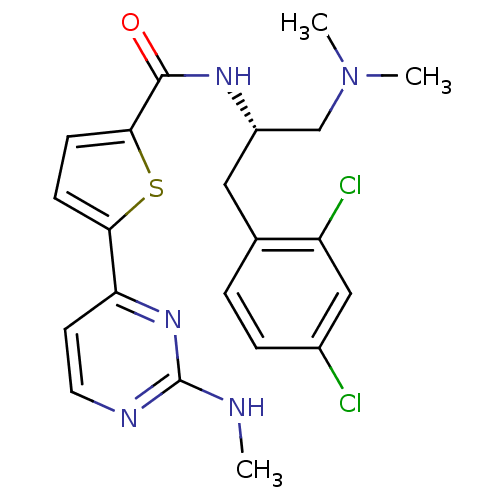
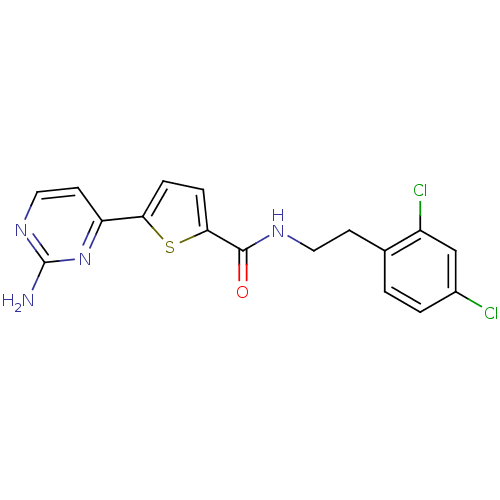
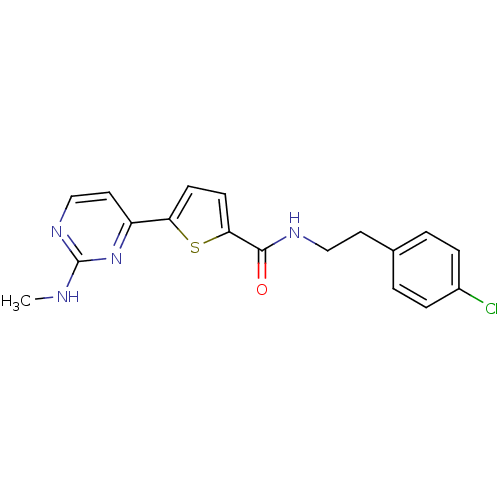
TargetCalcium/calmodulin-dependent protein kinase type II subunit alpha(Homo sapiens (Human))
Chiron
Curated by ChEMBL
Chiron
Curated by ChEMBL
Affinity DataIC50: >1nMAssay Description:Inhibition of CamK2alphaMore data for this Ligand-Target Pair
Affinity DataIC50: 2.60nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 4.80nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 11nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 27nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
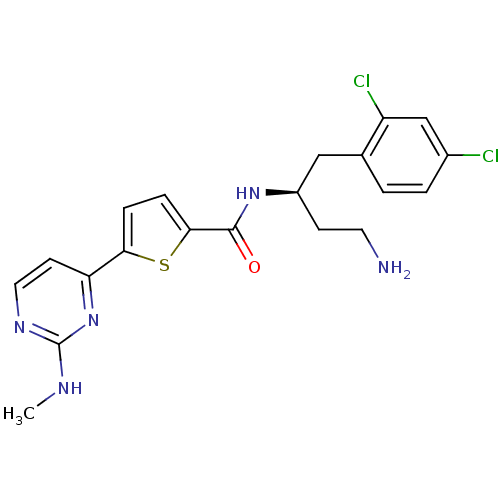
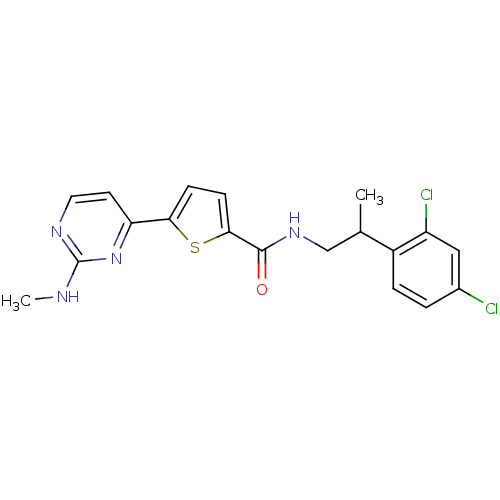
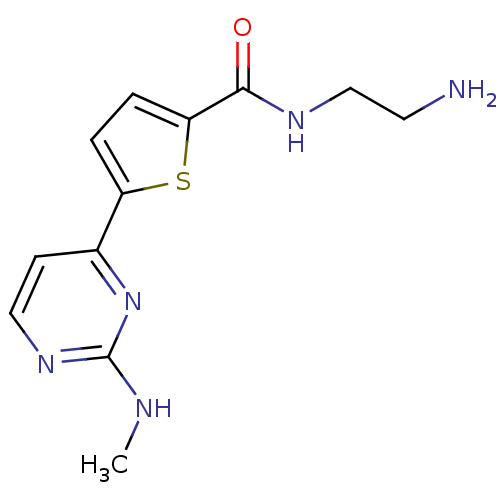
TargetRAF proto-oncogene serine/threonine-protein kinase(Homo sapiens (Human))
Chiron
Curated by ChEMBL
Chiron
Curated by ChEMBL
Affinity DataIC50: 240nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 280nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 610nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 660nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 820nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 990nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 1.10E+3nMAssay Description:Inhibition of MetMore data for this Ligand-Target Pair
Affinity DataIC50: 1.10E+3nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 1.30E+3nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 1.50E+3nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 2.10E+3nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 2.30E+3nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 2.40E+3nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 2.60E+3nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 2.80E+3nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 2.90E+3nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 4.70E+3nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 4.90E+3nMAssay Description:Inhibition of ERK1More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 5.20E+3nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 5.40E+3nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 7.00E+3nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 7.40E+3nMAssay Description:Inhibition of AKT3 in presence of 0.2 uM ATPMore data for this Ligand-Target Pair
TargetCalcium/calmodulin-dependent protein kinase type II subunit alpha(Homo sapiens (Human))
Chiron
Curated by ChEMBL
Chiron
Curated by ChEMBL
Affinity DataIC50: 8.00E+3nMAssay Description:Inhibition of CamK2alphaMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of EGFRMore data for this Ligand-Target Pair
TargetRAF proto-oncogene serine/threonine-protein kinase(Homo sapiens (Human))
Chiron
Curated by ChEMBL
Chiron
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of RAF1More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of GSK3More data for this Ligand-Target Pair

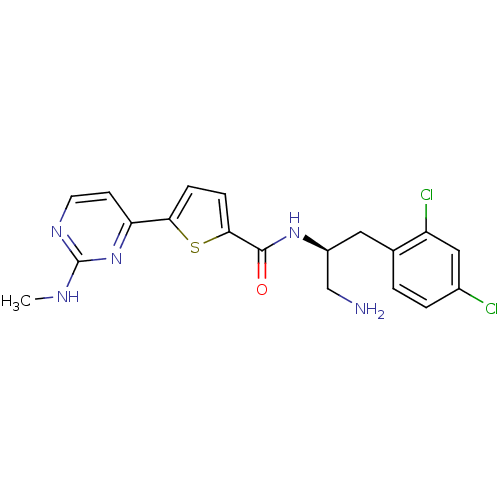
 3D Structure (crystal)
3D Structure (crystal)