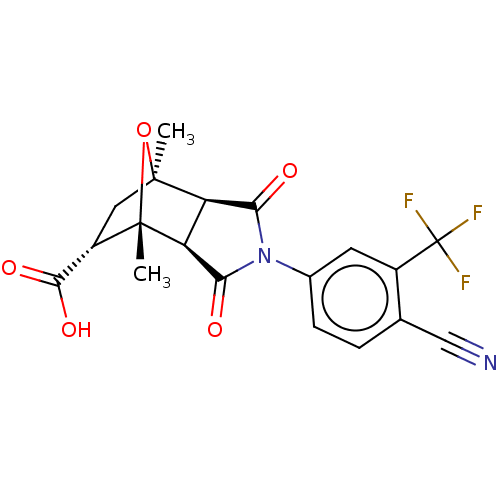
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 1nMAssay Description:Displacement of [3H]-DHT from androgen receptor in human MDA-MB-453 cells after 90 mins by TopCount analysisMore data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 1.70nMAssay Description:Displacement of [3H]-DHT from androgen receptor in human MDA-MB-453 cells after 90 mins by TopCount analysisMore data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 2nMAssay Description:Displacement of [3H]-DHT from androgen receptor in human MDA-MB-453 cells after 90 mins by TopCount analysisMore data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 3nMAssay Description:Displacement of [3H]-DHT from androgen receptor in human MDA-MB-453 cells after 90 mins by TopCount analysisMore data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 7nMAssay Description:Displacement of [3H]-DHT from androgen receptor in human MDA-MB-453 cells after 90 mins by TopCount analysisMore data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 8nMAssay Description:Displacement of [3H]-DHT from androgen receptor in human MDA-MB-453 cells after 90 mins by TopCount analysisMore data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 10nMAssay Description:Displacement of [3H]-DHT from androgen receptor in human MDA-MB-453 cells after 90 mins by TopCount analysisMore data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 12nMAssay Description:Displacement of [3H]-DHT from androgen receptor in human MDA-MB-453 cells after 90 mins by TopCount analysisMore data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 23nMAssay Description:Displacement of [3H]-DHT from androgen receptor in human MDA-MB-453 cells after 90 mins by TopCount analysisMore data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 31nMAssay Description:Displacement of [3H]-DHT from androgen receptor in human MDA-MB-453 cells after 90 mins by TopCount analysisMore data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 31nMAssay Description:Displacement of [3H]-DHT from androgen receptor in human MDA-MB-453 cells after 90 mins by TopCount analysisMore data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 50nMAssay Description:Displacement of [3H]-DHT from androgen receptor in human MDA-MB-453 cells after 90 mins by TopCount analysisMore data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 50nMAssay Description:Displacement of [3H]-DHT from androgen receptor in human MDA-MB-453 cells after 90 mins by TopCount analysisMore data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 64nMAssay Description:Displacement of [3H]-DHT from androgen receptor in human MDA-MB-453 cells after 90 mins by TopCount analysisMore data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 1.60E+3nMAssay Description:Displacement of [3H]-DHT from androgen receptor in human MDA-MB-453 cells after 90 mins by TopCount analysisMore data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 1nMAssay Description:Antagonist activity at androgen receptor in human MDA-MB-453 cells assessed as inhibition of DHT-induced PSA expression by alkaline phosphatase repor...More data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 6nMAssay Description:Antagonist activity at androgen receptor in human MDA-MB-453 cells assessed as inhibition of DHT-induced PSA expression by alkaline phosphatase repor...More data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 7nMAssay Description:Antagonist activity at androgen receptor in human MDA-MB-453 cells assessed as inhibition of DHT-induced PSA expression by alkaline phosphatase repor...More data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Antagonist activity at androgen receptor in human MDA-MB-453 cells assessed as inhibition of DHT-induced PSA expression by alkaline phosphatase repor...More data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Antagonist activity at androgen receptor in human MDA-MB-453 cells assessed as inhibition of DHT-induced PSA expression by alkaline phosphatase repor...More data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 16nMAssay Description:Antagonist activity at androgen receptor in human MDA-MB-453 cells assessed as inhibition of DHT-induced PSA expression by alkaline phosphatase repor...More data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 20nMAssay Description:Antagonist activity at androgen receptor in human MDA-MB-453 cells assessed as inhibition of DHT-induced PSA expression by alkaline phosphatase repor...More data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 22nMAssay Description:Antagonist activity at androgen receptor in human MDA-MB-453 cells assessed as inhibition of DHT-induced PSA expression by alkaline phosphatase repor...More data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 23nMAssay Description:Antagonist activity at androgen receptor in human MDA-MB-453 cells assessed as inhibition of DHT-induced PSA expression by alkaline phosphatase repor...More data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 34nMAssay Description:Antagonist activity at androgen receptor in human MDA-MB-453 cells assessed as inhibition of DHT-induced PSA expression by alkaline phosphatase repor...More data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 34nMAssay Description:Antagonist activity at androgen receptor in human MDA-MB-453 cells assessed as inhibition of DHT-induced PSA expression by alkaline phosphatase repor...More data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 60nMAssay Description:Antagonist activity at androgen receptor in human MDA-MB-453 cells assessed as inhibition of DHT-induced PSA expression by alkaline phosphatase repor...More data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 60nMAssay Description:Antagonist activity at androgen receptor in human MDA-MB-453 cells assessed as inhibition of DHT-induced PSA expression by alkaline phosphatase repor...More data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 173nMAssay Description:Antagonist activity at androgen receptor in human MDA-MB-453 cells assessed as inhibition of DHT-induced PSA expression by alkaline phosphatase repor...More data for this Ligand-Target Pair
TargetAndrogen receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: >5.00E+3nMAssay Description:Antagonist activity at androgen receptor in human MDA-MB-453 cells assessed as inhibition of DHT-induced PSA expression by alkaline phosphatase repor...More data for this Ligand-Target Pair
TargetCytochrome P450 1A2(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: >4.00E+4nMAssay Description:Inhibition of human CYP1A2More data for this Ligand-Target Pair
TargetCytochrome P450 2B6(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: >4.00E+4nMAssay Description:Inhibition of human CYP2B6More data for this Ligand-Target Pair
TargetCytochrome P450 2C8(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: >4.00E+4nMAssay Description:Inhibition of human CYP2C8More data for this Ligand-Target Pair
TargetCytochrome P450 2C9(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: >4.00E+4nMAssay Description:Inhibition of human CYP2C9More data for this Ligand-Target Pair
TargetCytochrome P450 2D6(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: >4.00E+4nMAssay Description:Inhibition of human CYP2D6More data for this Ligand-Target Pair
TargetCytochrome P450 3A4(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: >4.00E+4nMAssay Description:Inhibition of human CYP3A4More data for this Ligand-Target Pair
TargetNuclear receptor subfamily 1 group I member 2(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataEC50: >5.00E+4nMAssay Description:Transactivation of human PXRMore data for this Ligand-Target Pair



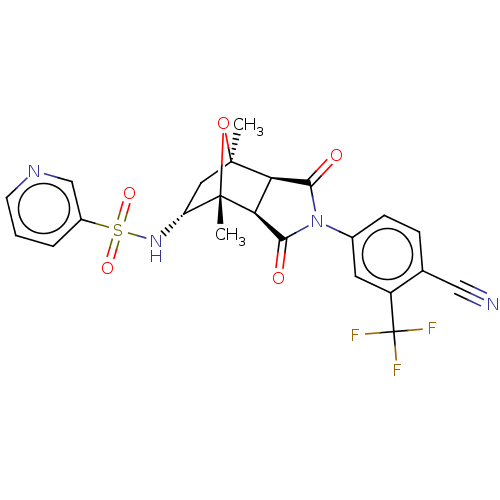
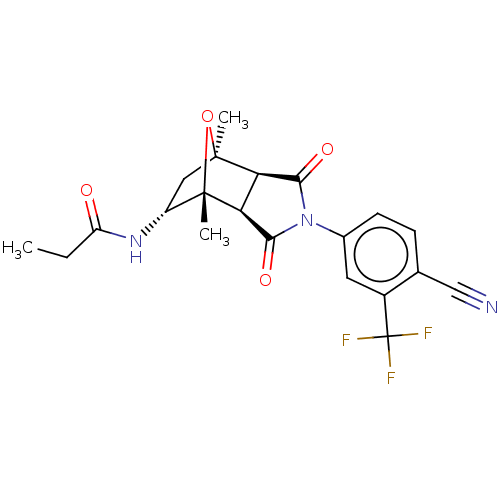



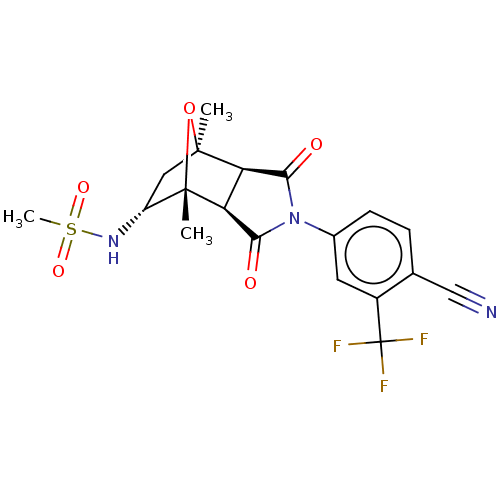
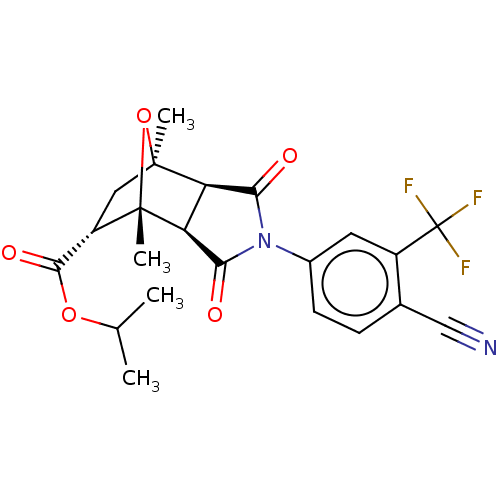


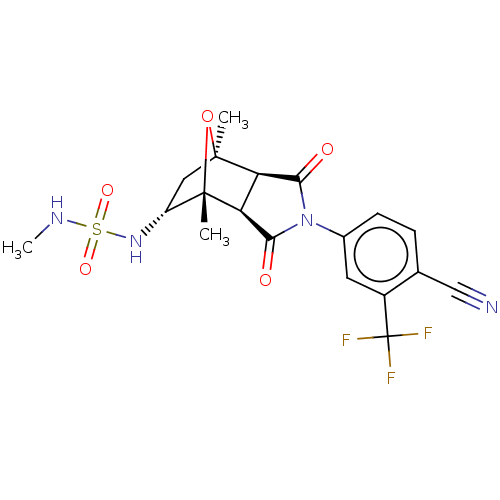
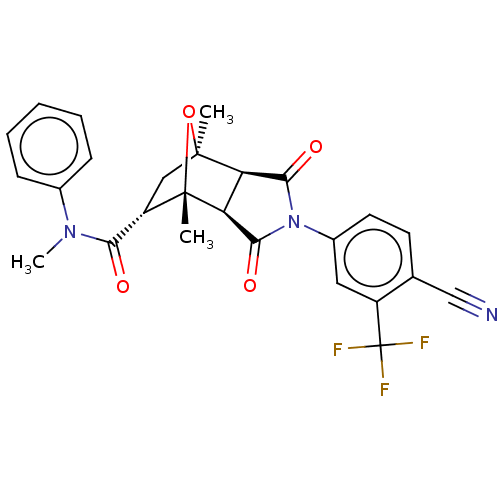
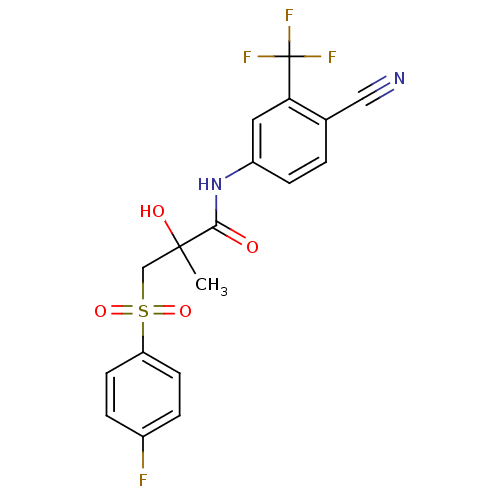
 3D Structure (crystal)
3D Structure (crystal)