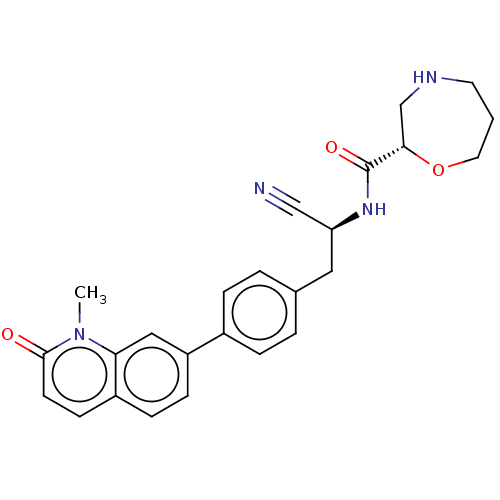
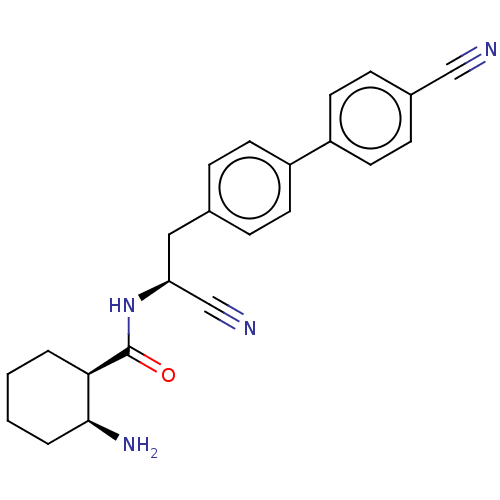
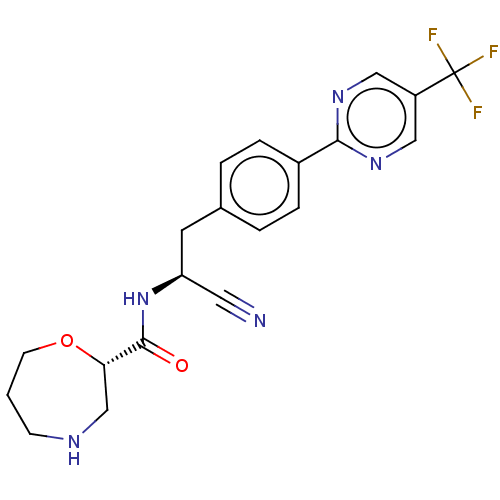
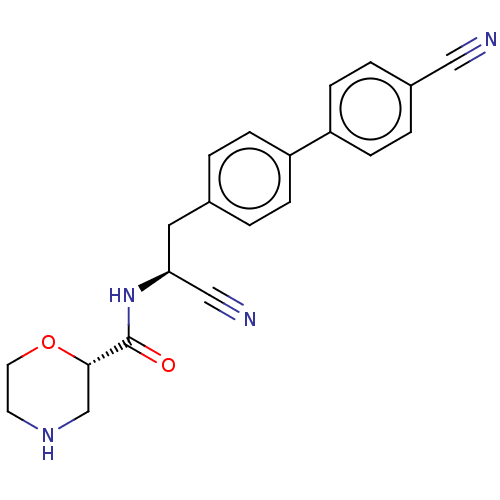
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 0.251nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 0.794nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 1.60nMAssay Description:Inhibition of DPP1 in human U937 cells using Gly-Phe-AFC as substrate preincubated for 60 mins followed by substrate addition by fluorescence assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 2.5nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 3.20nMAssay Description:Inhibition of DPP1 in human U937 cells using Gly-Phe-AFC as substrate preincubated for 60 mins followed by substrate addition by fluorescence assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 4nMAssay Description:Inhibition of DPP1 in human U937 cells using Gly-Phe-AFC as substrate preincubated for 60 mins followed by substrate addition by fluorescence assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 4nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 5nMAssay Description:Inhibition of DPP1 in human U937 cells using Gly-Phe-AFC as substrate preincubated for 60 mins followed by substrate addition by fluorescence assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 5nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 6.30nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 6.30nMAssay Description:Inhibition of DPP1 in human U937 cells using Gly-Phe-AFC as substrate preincubated for 60 mins followed by substrate addition by fluorescence assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 7.90nMAssay Description:Inhibition of DPP1 in human U937 cells using Gly-Phe-AFC as substrate preincubated for 60 mins followed by substrate addition by fluorescence assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 7.90nMAssay Description:Inhibition of DPP1 in human U937 cells using Gly-Phe-AFC as substrate preincubated for 60 mins followed by substrate addition by fluorescence assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 13nMAssay Description:Inhibition of DPP1 in human U937 cells using Gly-Phe-AFC as substrate preincubated for 60 mins followed by substrate addition by fluorescence assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 13nMAssay Description:Inhibition of DPP1 in human U937 cells using Gly-Phe-AFC as substrate preincubated for 60 mins followed by substrate addition by fluorescence assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 13nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 13nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 16nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 16nMAssay Description:Inhibition of DPP1 in human U937 cells using Gly-Phe-AFC as substrate preincubated for 60 mins followed by substrate addition by fluorescence assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 16nMAssay Description:Inhibition of DPP1 in human U937 cells using Gly-Phe-AFC as substrate preincubated for 60 mins followed by substrate addition by fluorescence assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Canis lupus familiaris)
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 16nMAssay Description:Inhibition of dog DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 16nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Rattus norvegicus)
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 20nMAssay Description:Inhibition of rat DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Mus musculus)
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 25nMAssay Description:Inhibition of mouse DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 25nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 32nMAssay Description:Inhibition of DPP1 in human U937 cells using Gly-Phe-AFC as substrate preincubated for 60 mins followed by substrate addition by fluorescence assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 32nMAssay Description:Inhibition of DPP1 in human U937 cells using Gly-Phe-AFC as substrate preincubated for 60 mins followed by substrate addition by fluorescence assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 40nMAssay Description:Inhibition of DPP1 in human U937 cells using Gly-Phe-AFC as substrate preincubated for 60 mins followed by substrate addition by fluorescence assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 40nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 40nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 40nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 40nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 62nMAssay Description:Inhibition of DPP1 in human primary bone marrow-derived CD34+ neutrophil progenitor cells assessed as inactivation of neutrophil elastase using metho...More data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 63nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 63nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 115nMAssay Description:Inhibition of DPP1 in human primary bone marrow-derived CD34+ neutrophil progenitor cells assessed as inactivation of cathepsin G using N-succinyl-Al...More data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 141nMAssay Description:Inhibition of DPP1 in human primary bone marrow-derived CD34+ neutrophil progenitor cellsMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 158nMAssay Description:Inhibition of DPP1 in human U937 cells using Gly-Phe-AFC as substrate preincubated for 60 mins followed by substrate addition by fluorescence assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 200nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 200nMAssay Description:Inhibition of DPP1 in human U937 cells using Gly-Phe-AFC as substrate preincubated for 60 mins followed by substrate addition by fluorescence assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 209nMAssay Description:Inhibition of DPP1 in human primary bone marrow-derived CD34+ neutrophil progenitor cells assessed as inactivation of proteinase 3 using aminobenzoyl...More data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 316nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 398nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 398nMAssay Description:Inhibition of DPP1 in human U937 cells using Gly-Phe-AFC as substrate preincubated for 60 mins followed by substrate addition by fluorescence assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 501nMAssay Description:Inhibition of DPP1 in human U937 cells using Gly-Phe-AFC as substrate preincubated for 60 mins followed by substrate addition by fluorescence assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 501nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 501nMAssay Description:Inhibition of DPP1 in human U937 cells using Gly-Phe-AFC as substrate preincubated for 60 mins followed by substrate addition by fluorescence assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 1(Homo sapiens (Human))
Charles River Discovery Research Services
Curated by ChEMBL
Charles River Discovery Research Services
Curated by ChEMBL
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibition of human recombinant DPP1 using Gly-Arg-AMC as substrate preincubated for 30 mins followed by substrate addition by fluorometric assayMore data for this Ligand-Target Pair