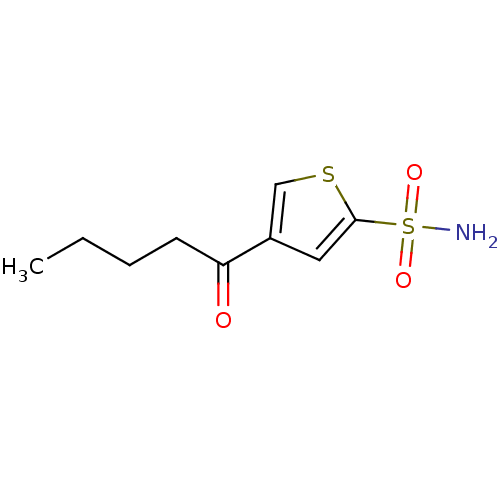
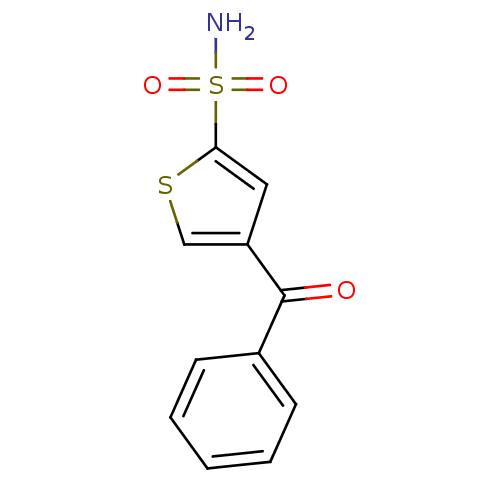
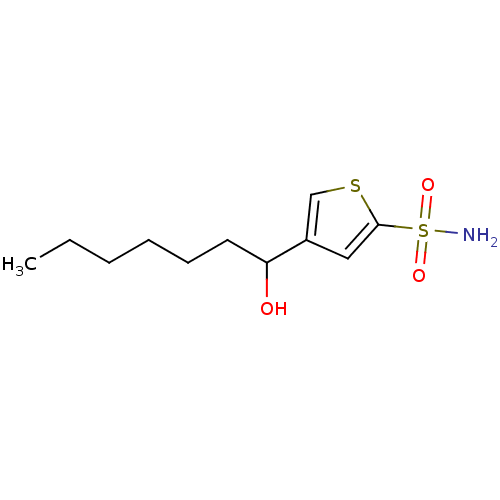
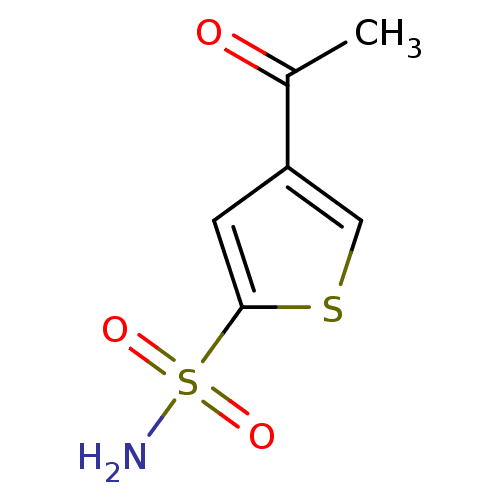
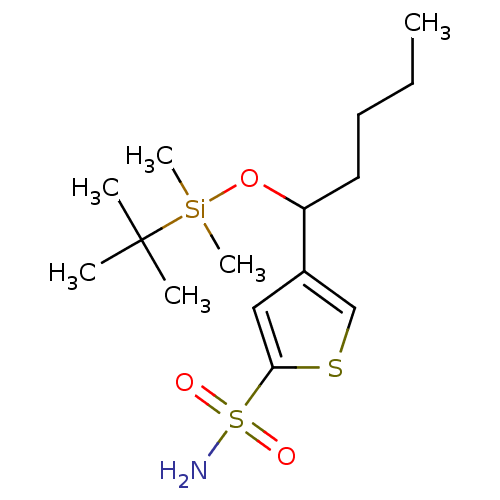
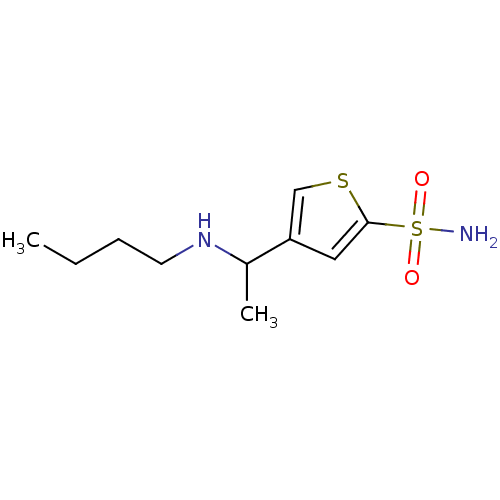
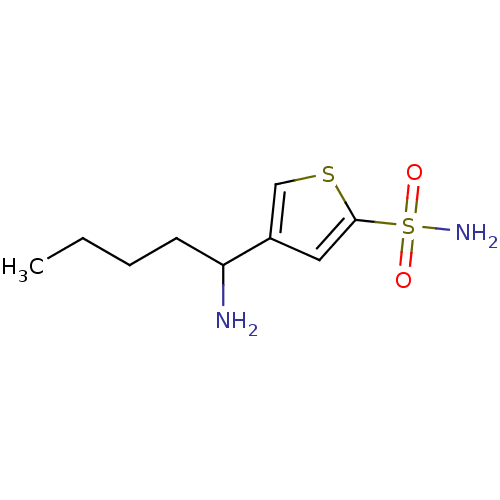
Affinity DataIC50: 2nMAssay Description:Activity against human carbonic anhydrase IIMore data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:Activity against human carbonic anhydrase IIMore data for this Ligand-Target Pair
Affinity DataIC50: 4.70nMAssay Description:Activity against human carbonic anhydrase IIMore data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Activity against human carbonic anhydrase IIMore data for this Ligand-Target Pair
Affinity DataIC50: 5.80nMAssay Description:Activity against human carbonic anhydrase IIMore data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Activity against human carbonic anhydrase IIMore data for this Ligand-Target Pair
Affinity DataIC50: 6.30nMAssay Description:Activity against human carbonic anhydrase IIMore data for this Ligand-Target Pair
Affinity DataIC50: 6.80nMAssay Description:Activity against human carbonic anhydrase IIMore data for this Ligand-Target Pair
Affinity DataIC50: 13nMAssay Description:Activity against human carbonic anhydrase IIMore data for this Ligand-Target Pair
Affinity DataIC50: 15nMAssay Description:Activity against human carbonic anhydrase IIMore data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:Activity against human carbonic anhydrase IIMore data for this Ligand-Target Pair
Affinity DataIC50: 160nMAssay Description:Activity against human carbonic anhydrase IIMore data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Activity against human carbonic anhydrase IIMore data for this Ligand-Target Pair
Affinity DataIC50: 590nMAssay Description:Activity against human carbonic anhydrase IIMore data for this Ligand-Target Pair
Affinity DataIC50: 626nMAssay Description:Activity against human carbonic anhydrase IIMore data for this Ligand-Target Pair

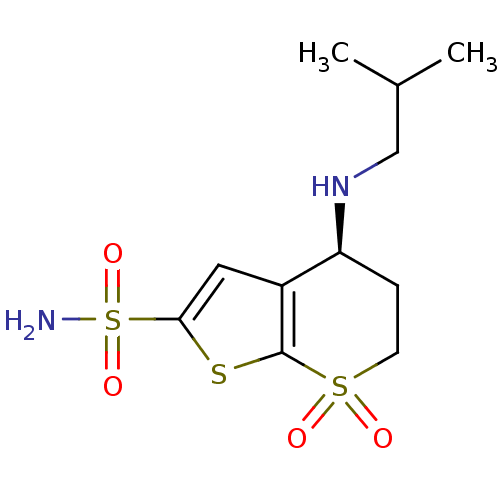

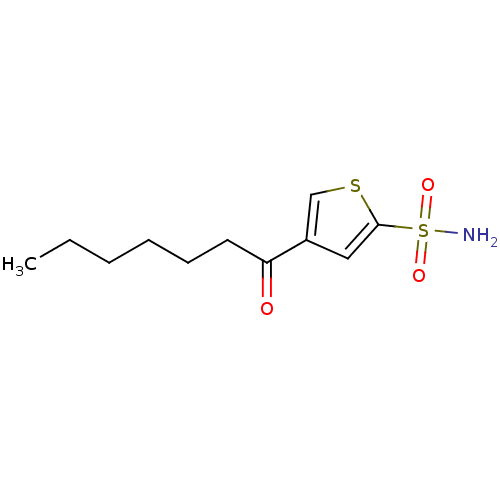
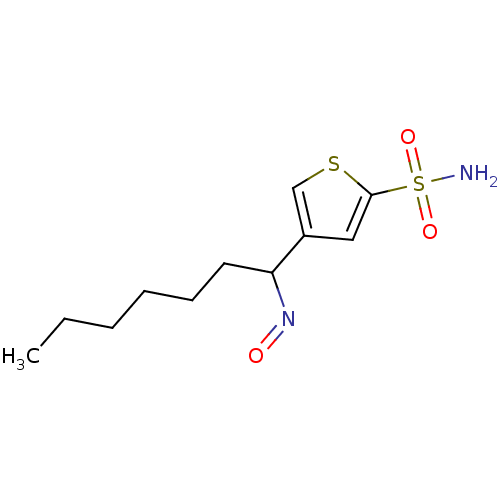

 3D Structure (crystal)
3D Structure (crystal)