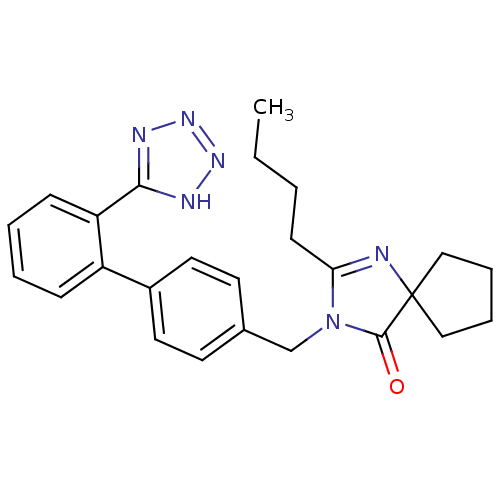
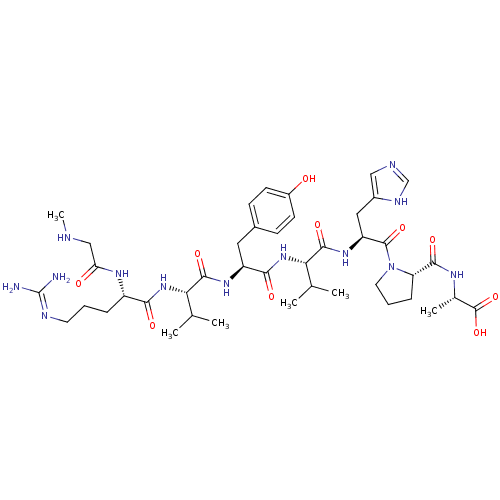
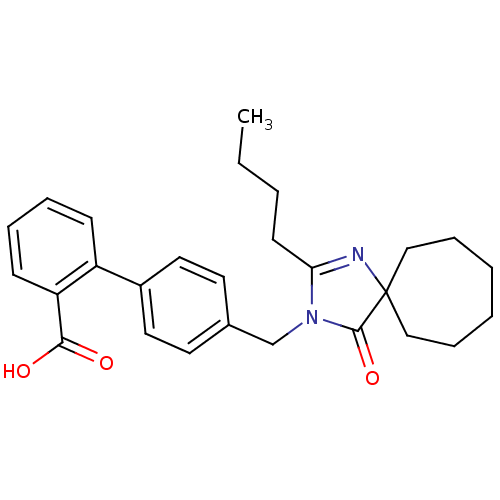
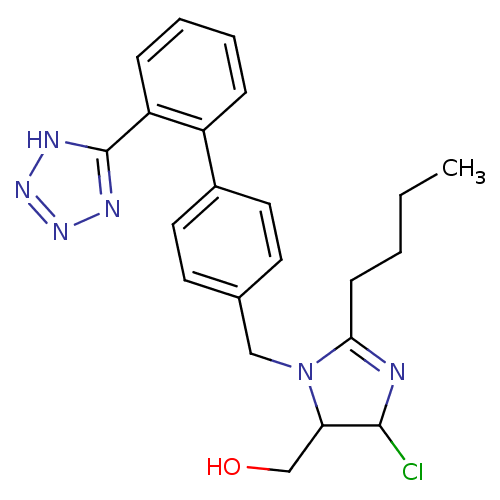
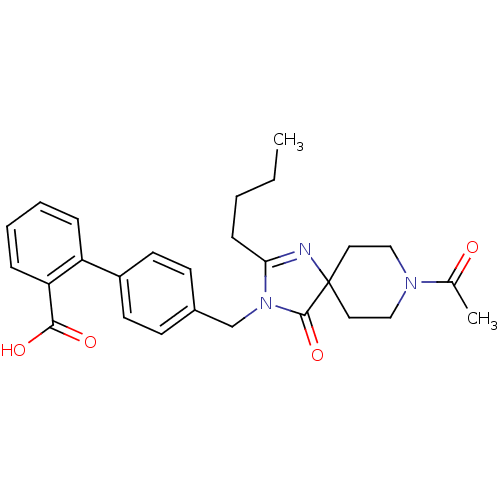
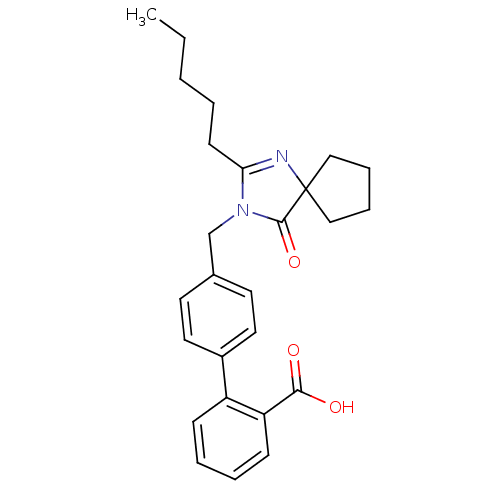
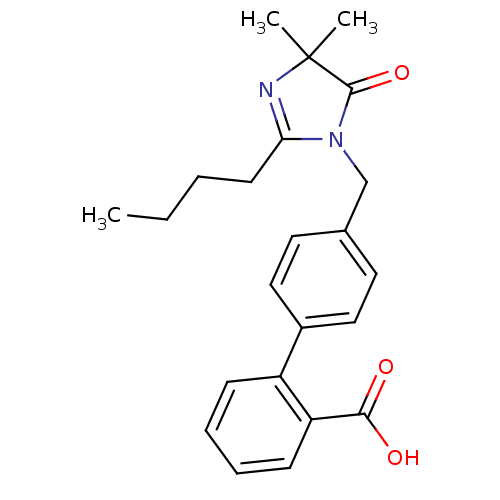
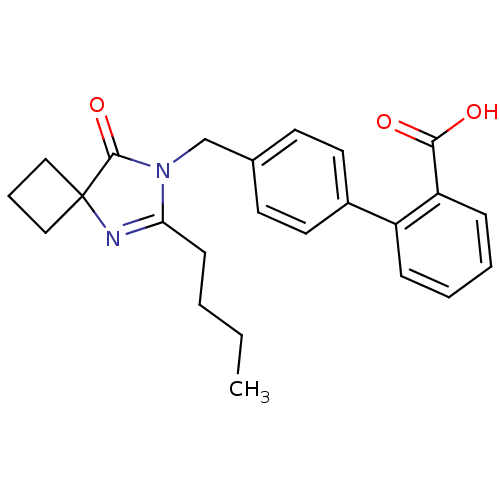
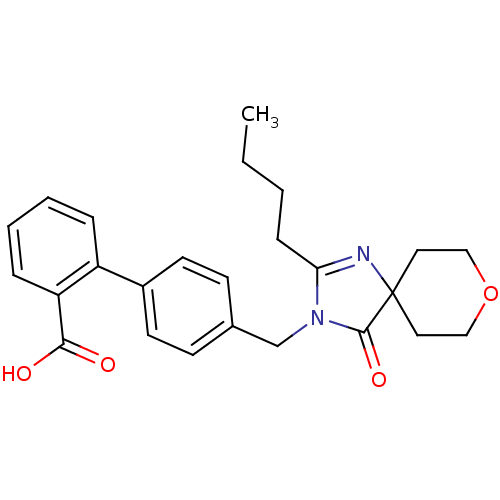
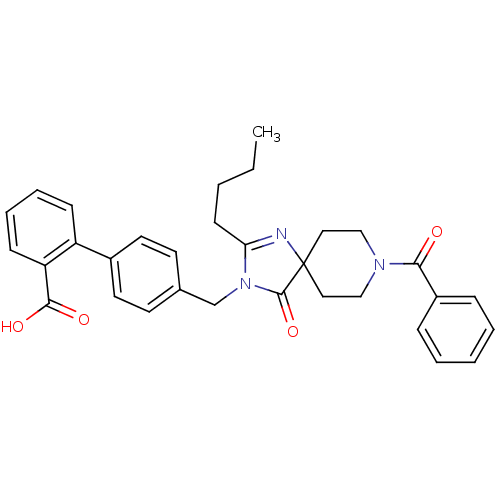
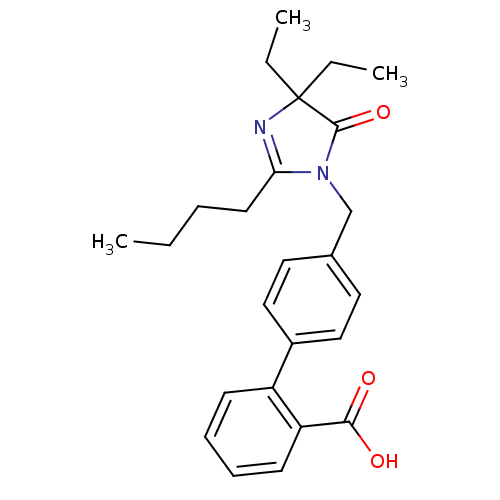
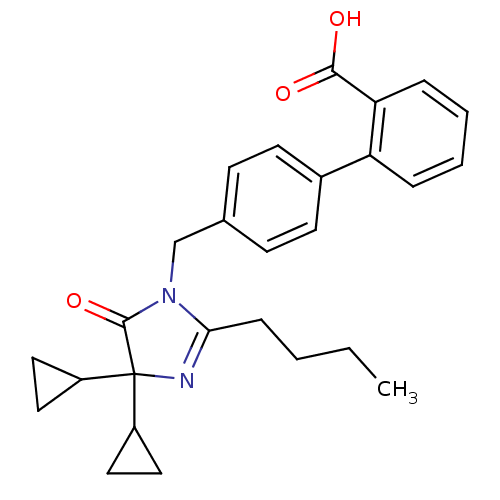
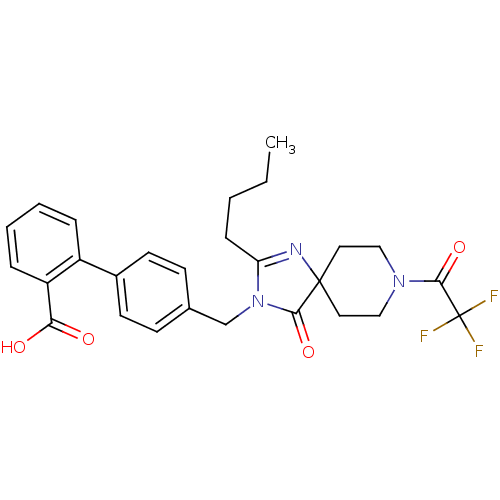
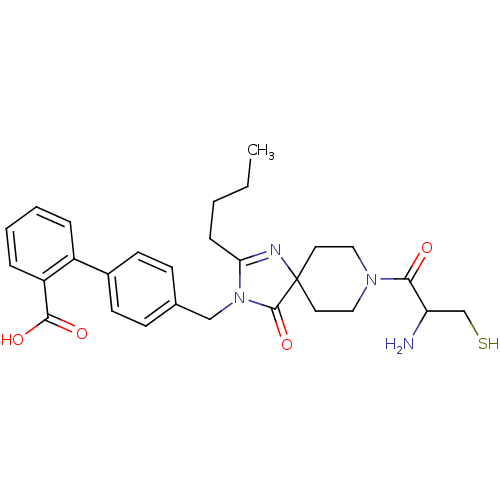
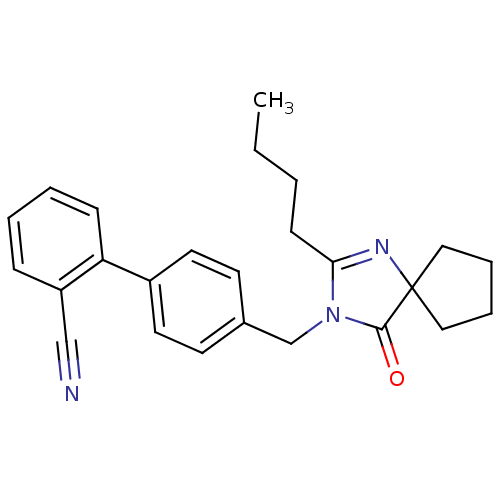
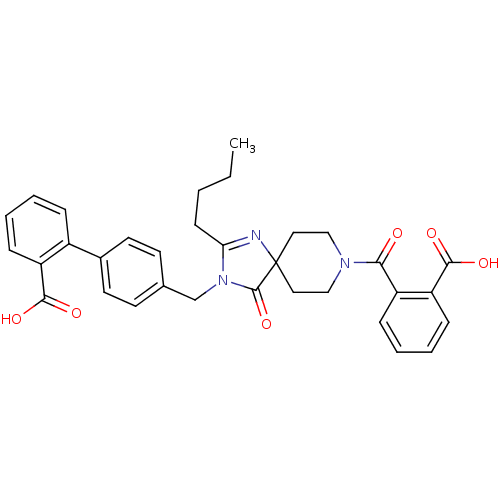
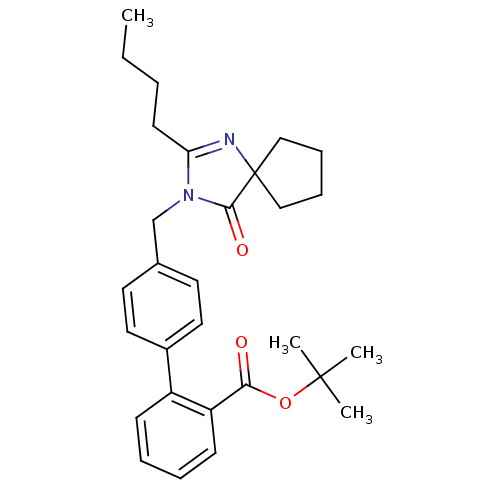
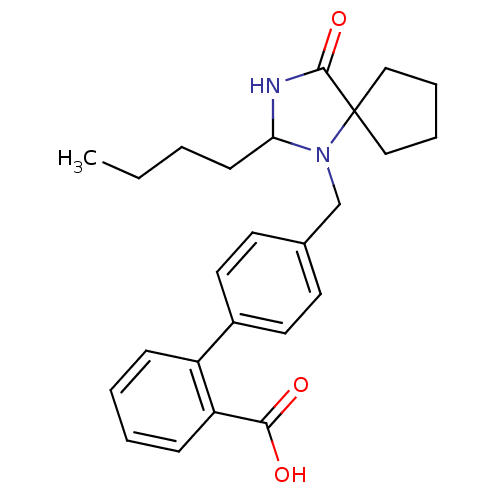
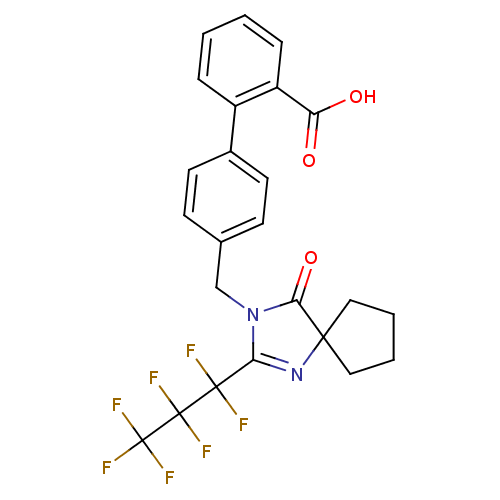
Affinity DataIC50: 1.30nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 2.40nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 7.20nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 8.70nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 14nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 18nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 24nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 27nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 35nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 38nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 47nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 49nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 56nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 59nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 60nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 62nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 66nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 80nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 80nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 110nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 140nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 190nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 220nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 250nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 260nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 260nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 350nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 410nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 440nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 480nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 660nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 730nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 800nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 890nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 950nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 1.70E+3nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 1.80E+3nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 1.80E+3nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 2.10E+3nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 2.50E+3nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 3.20E+3nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 3.30E+3nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 5.20E+3nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 7.00E+3nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Binding affinity for Angiotensin II receptor, type 1 measured by ability to displace [125I]- A II from its specific binding site in rat liver membran...More data for this Ligand-Target Pair