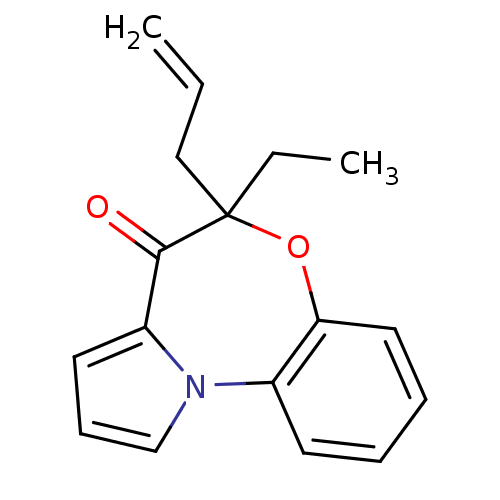
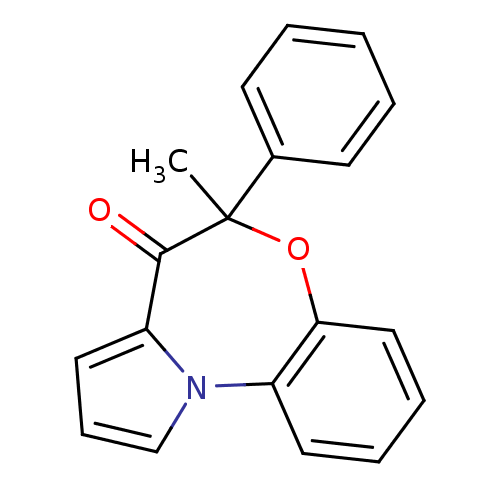
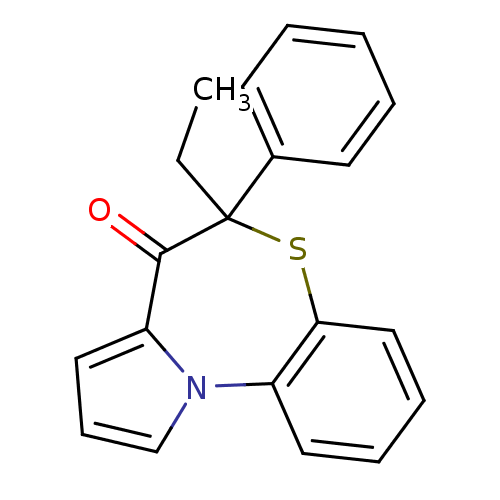
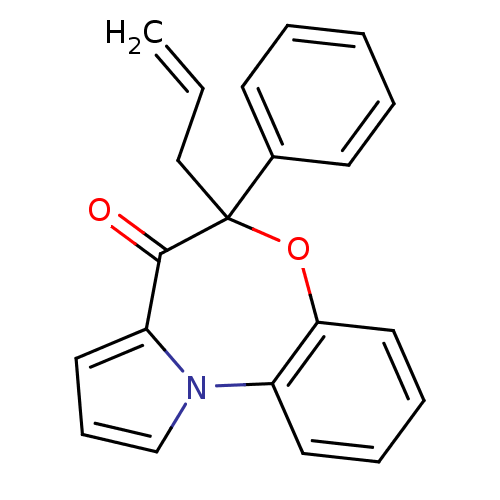
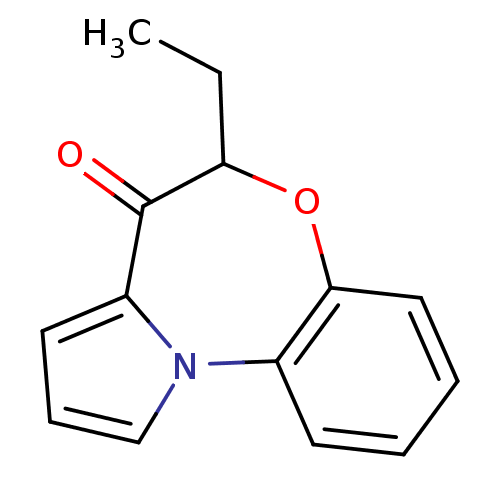
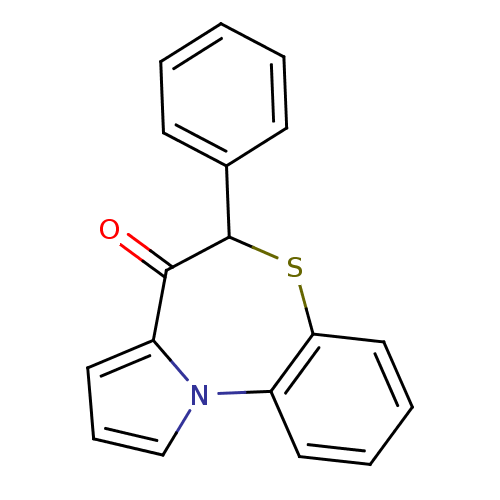
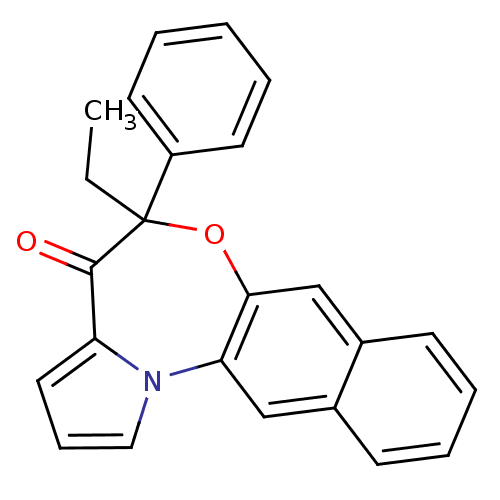
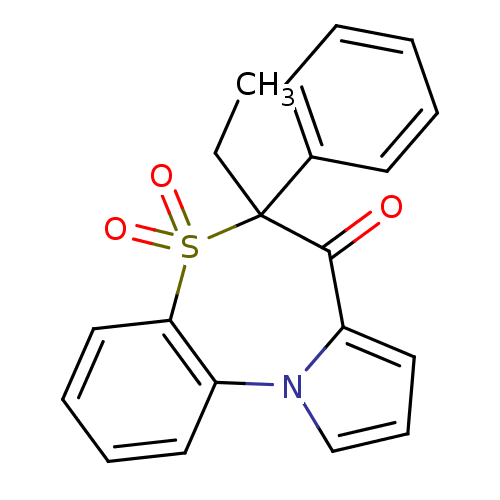
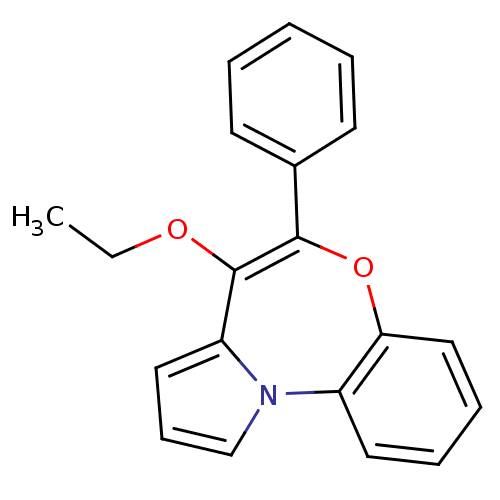
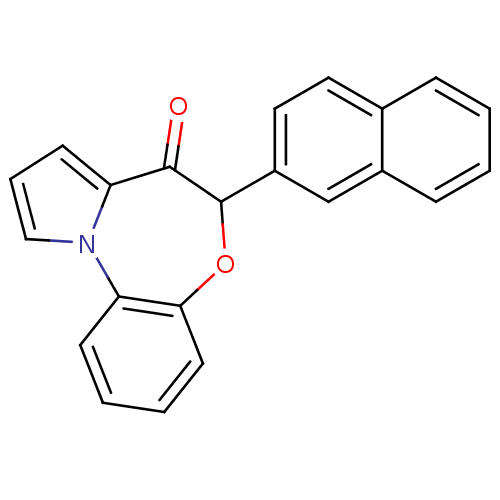
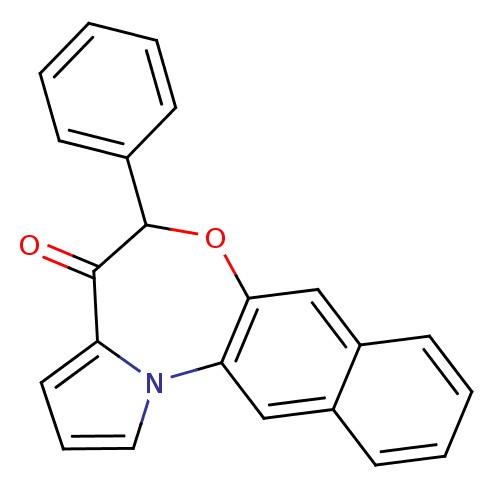
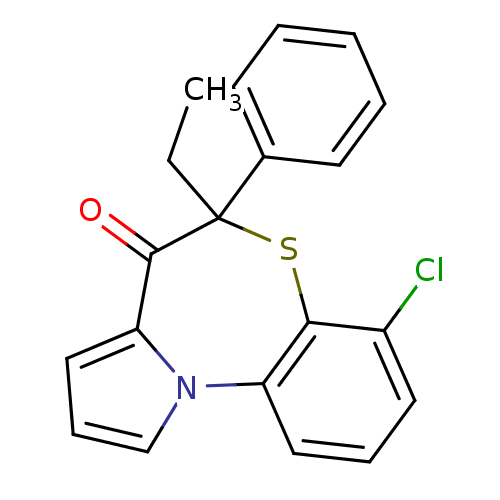
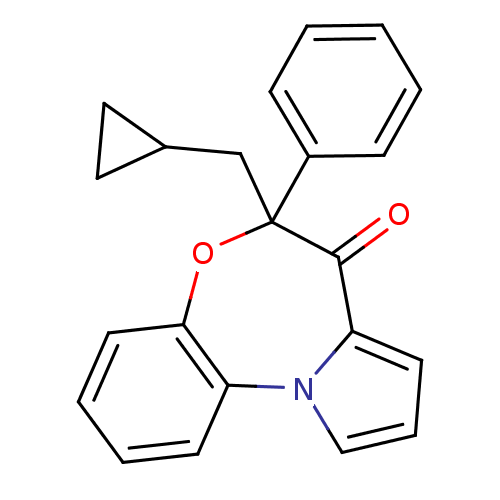
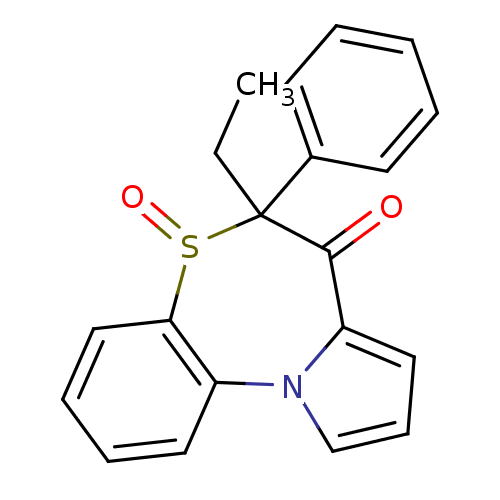
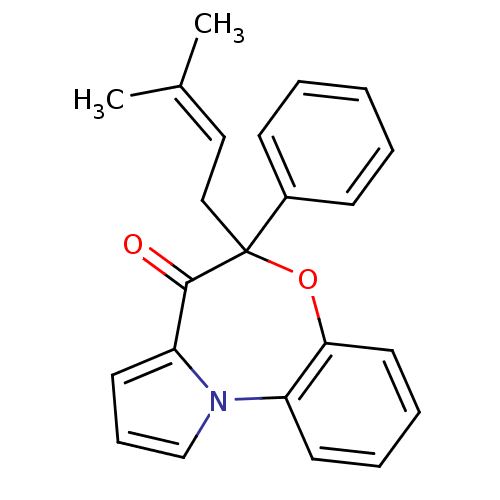
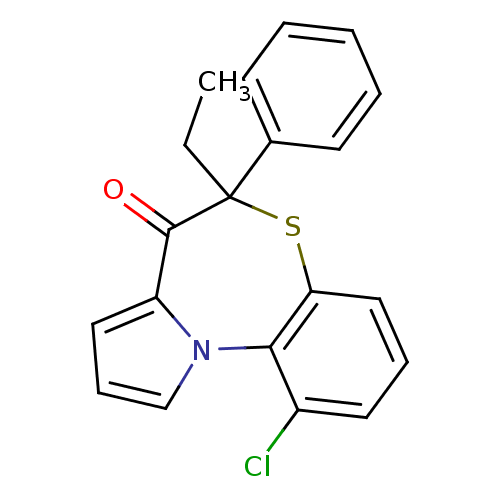
Affinity DataIC50: 150nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: 250nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activityMore data for this Ligand-Target Pair
Affinity DataIC50: 500nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: 500nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+3nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+3nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: 1.50E+4nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: 2.50E+4nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: 1.50E+5nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: >3.00E+5nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: >3.00E+5nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: >3.00E+5nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: >3.00E+5nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: >3.00E+5nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+5nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: >5.00E+5nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+5nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: >5.00E+5nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+5nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: 6.00E+5nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair
Affinity DataIC50: >6.00E+5nMAssay Description:Compound was tested for Inhibition of HIV-1 RT activity.More data for this Ligand-Target Pair

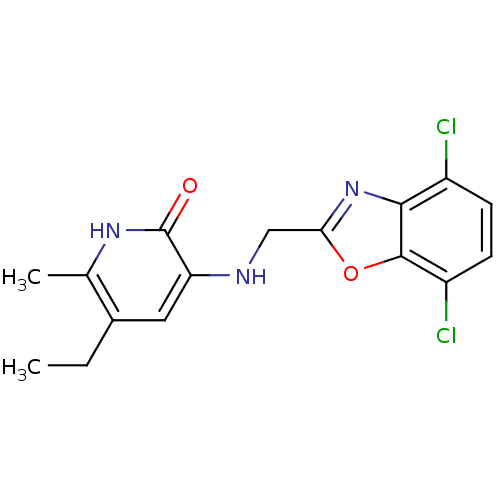
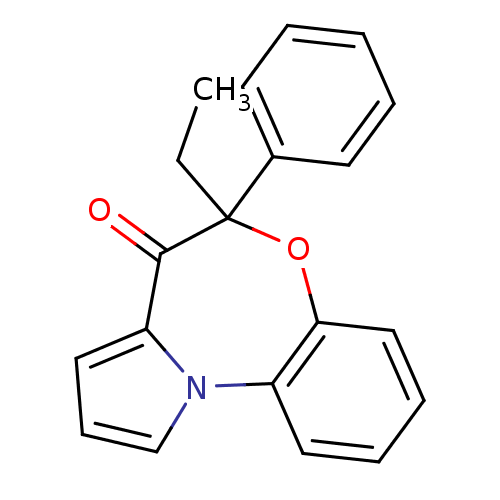
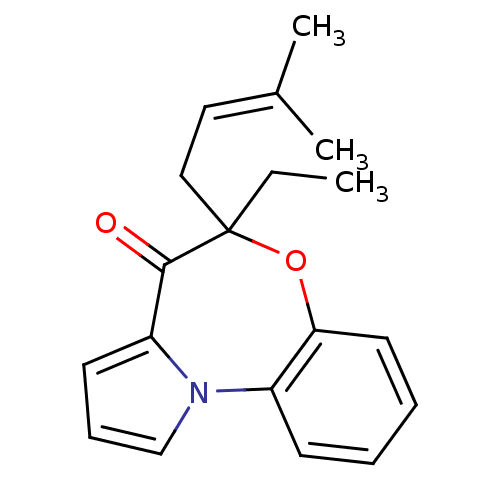
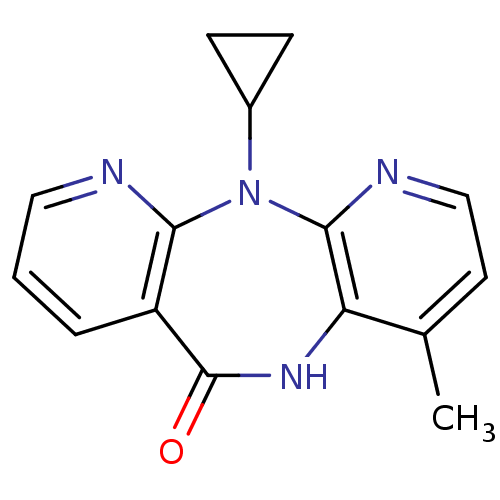
 3D Structure (crystal)
3D Structure (crystal)