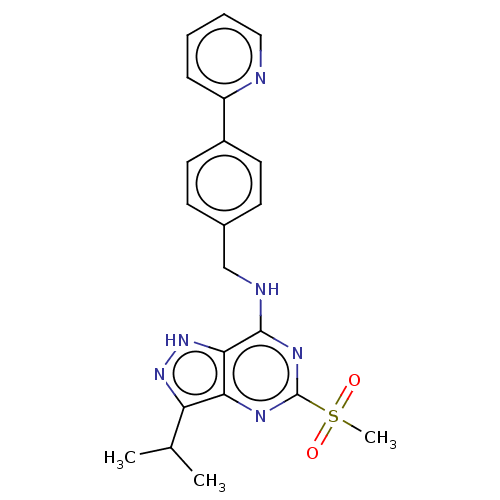
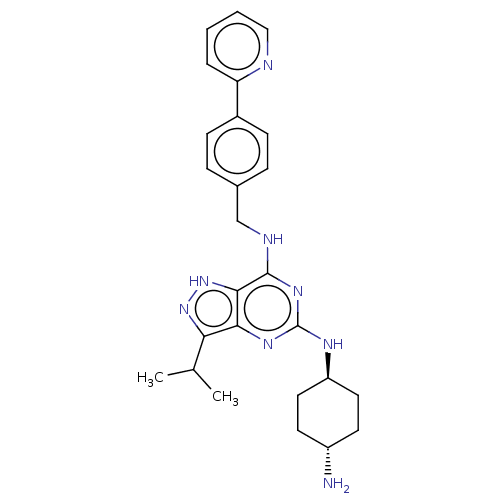
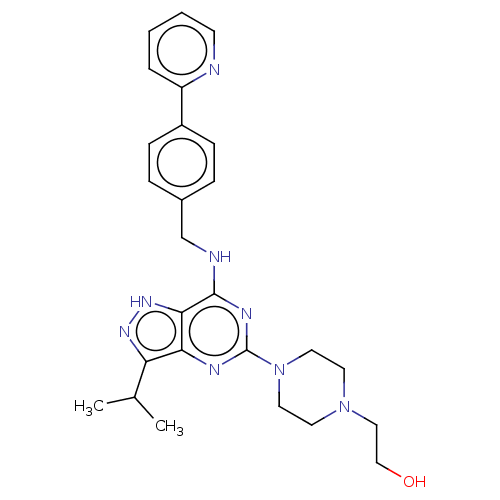
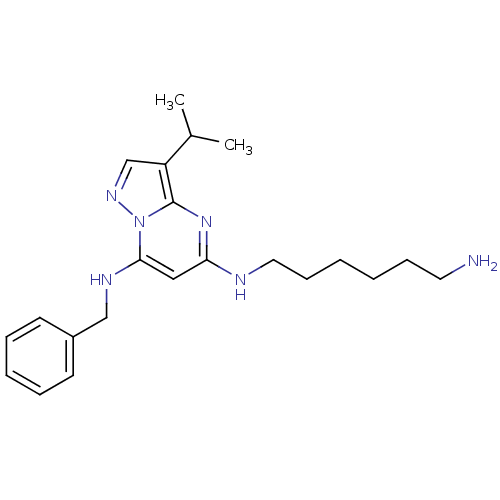
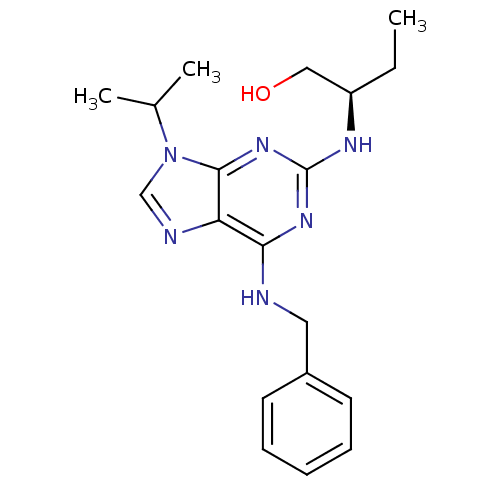
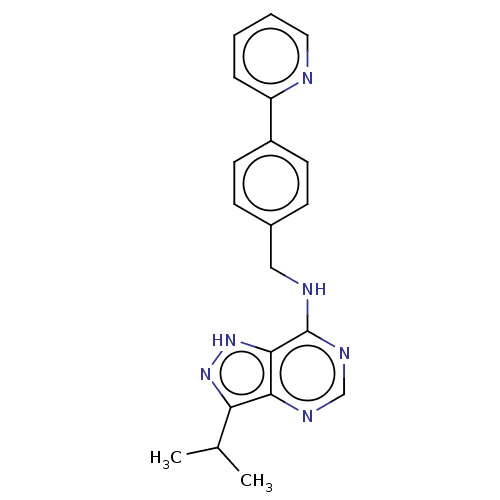
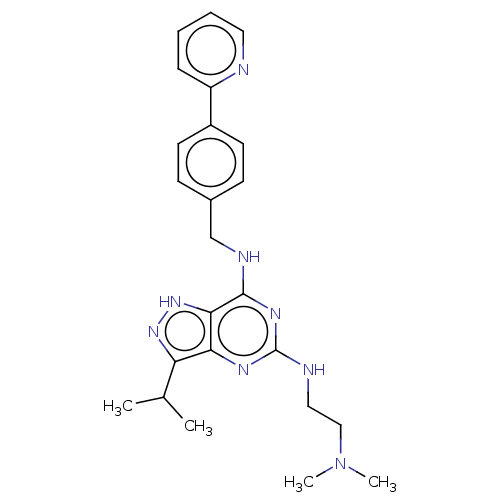
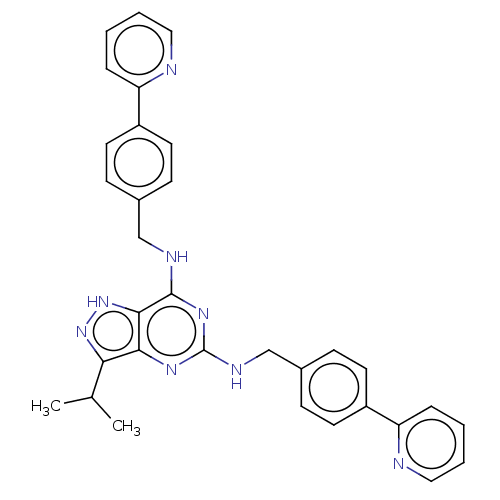
Affinity DataIC50: 1nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 2nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 8nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 9nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 9nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 12nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:Inhibition of CDK4 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 17nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 18nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 18nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 21nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 21nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 24nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 31nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 37nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 44nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 45nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 46nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 48nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 49nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 50nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 51nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 54nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 61nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 62nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 70nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 72nMAssay Description:Inhibition of CDK1 (unknown origin)More data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 96nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 114nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 119nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 127nMAssay Description:Inhibition of CDK4 (unknown origin)More data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 132nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 134nMAssay Description:Inhibition of CDK7 (unknown origin)More data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 158nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 165nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 178nMAssay Description:Inhibition of CDK9 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 179nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 183nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 197nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 225nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 229nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 240nMAssay Description:Inhibition of CDK9 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 422nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 467nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 777nMAssay Description:Inhibition of CDK1 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 892nMAssay Description:Inhibition of CDK9 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 1.07E+3nMAssay Description:Inhibition of CDK5 (unknown origin) using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair
TargetCyclin-dependent kinase 2/G1/S-specific cyclin-E1(Homo sapiens (Human))
Palack£
Curated by ChEMBL
Palack£
Curated by ChEMBL
Affinity DataIC50: 1.45E+3nMAssay Description:Inhibition of CDK2/Cyclin E (unknown origin) expressed in sf9 cells using histone H1 as substrate in presence of [gamma33P]-ATPMore data for this Ligand-Target Pair

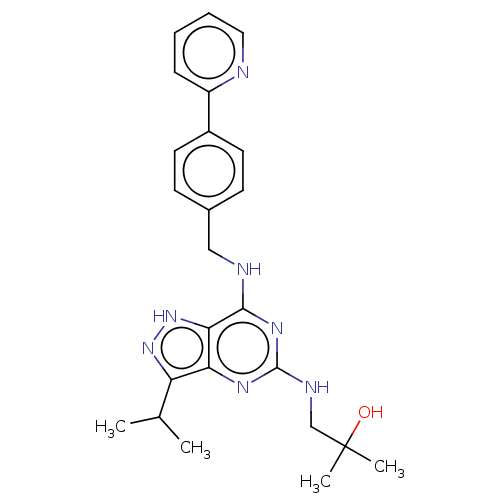
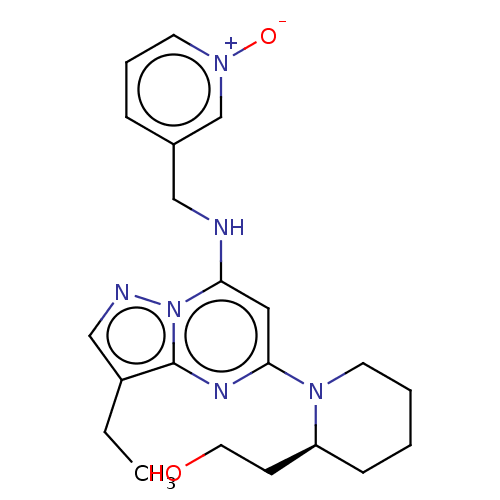


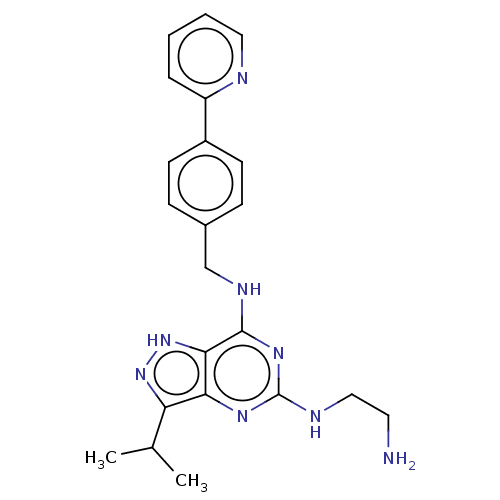

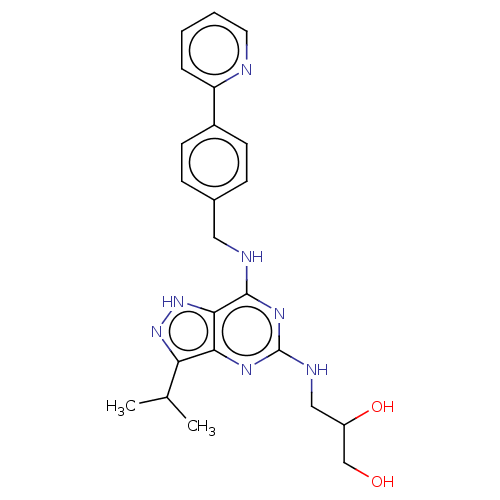




 3D Structure (crystal)
3D Structure (crystal)