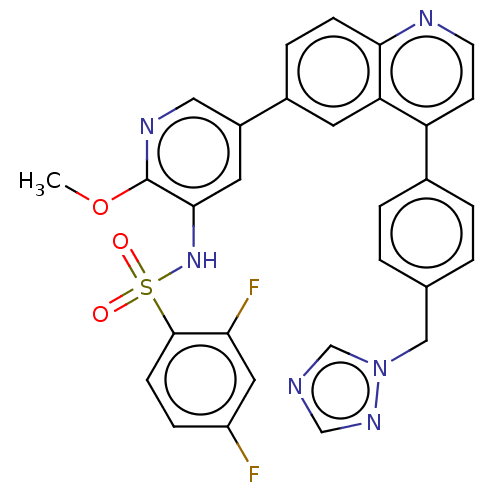
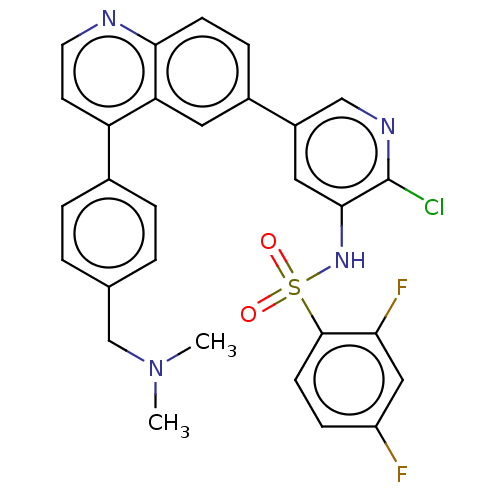
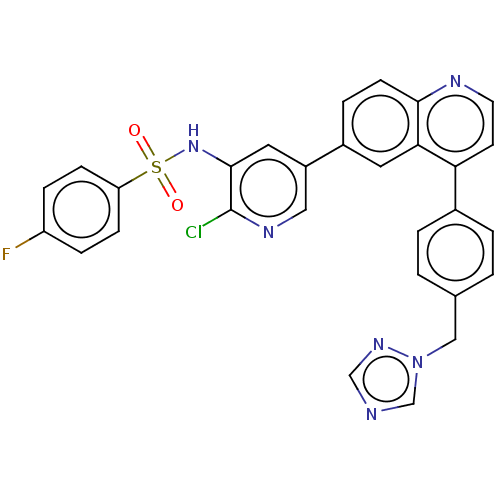
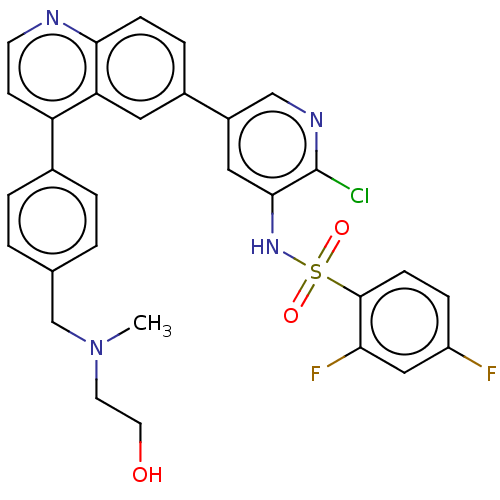
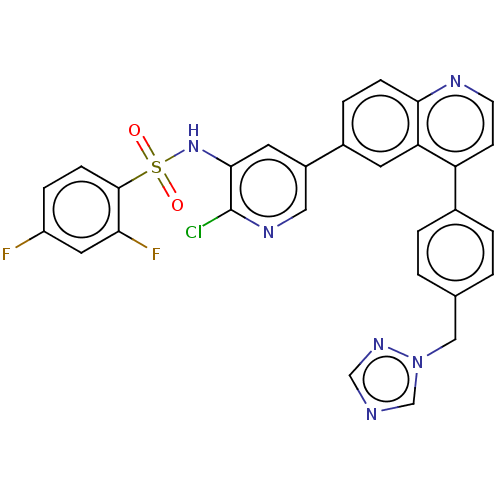
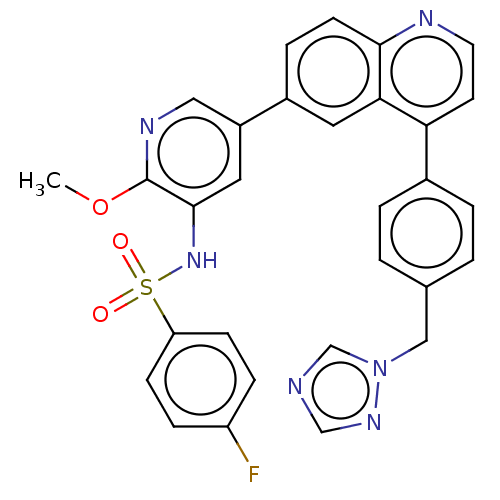
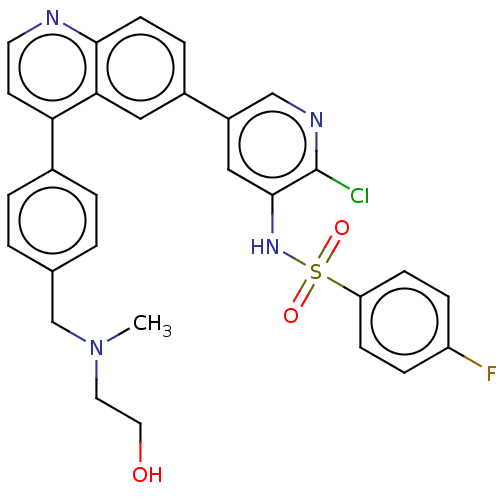
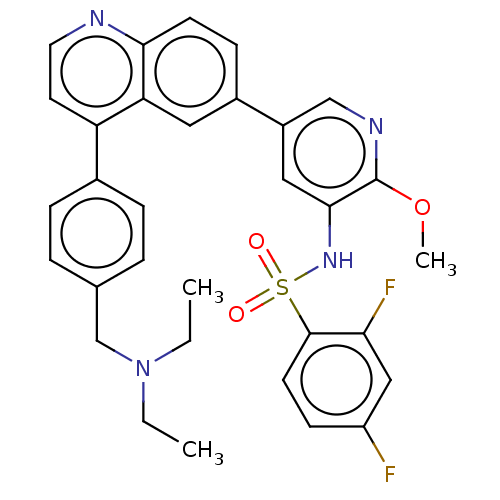
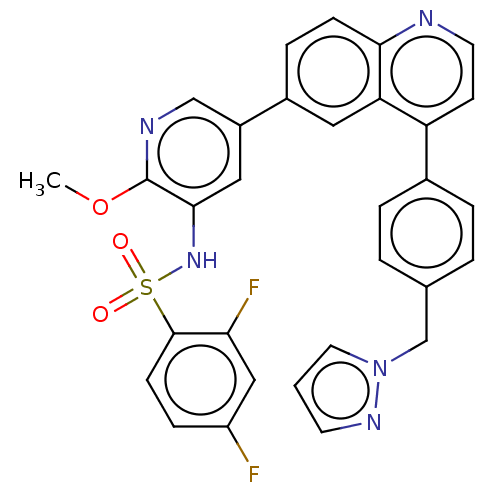
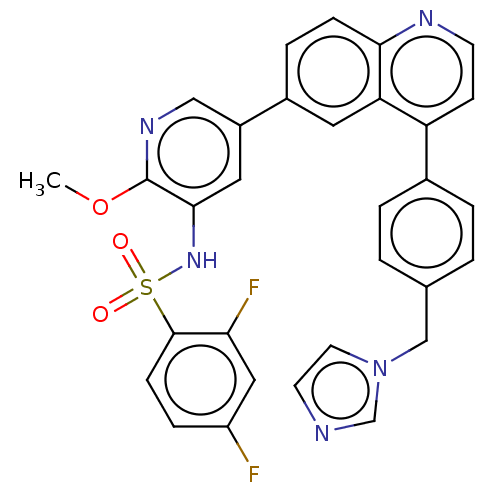
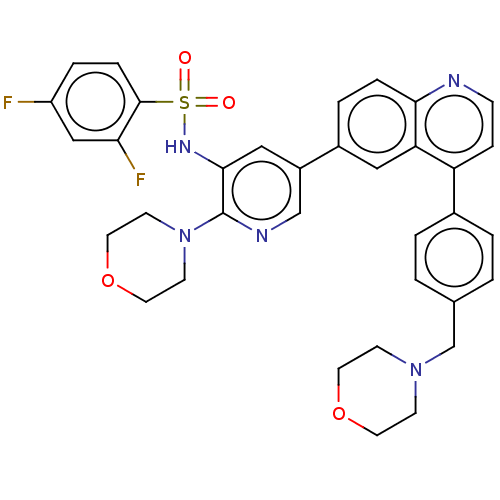
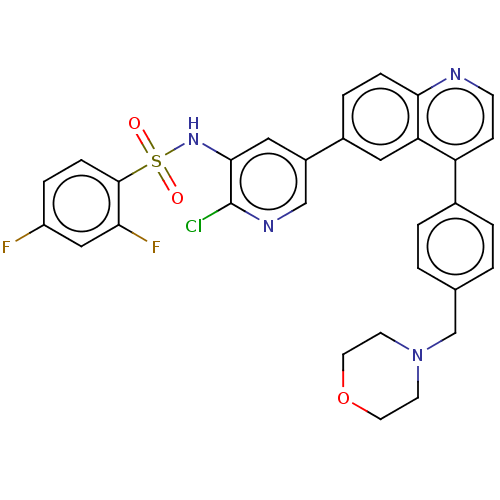
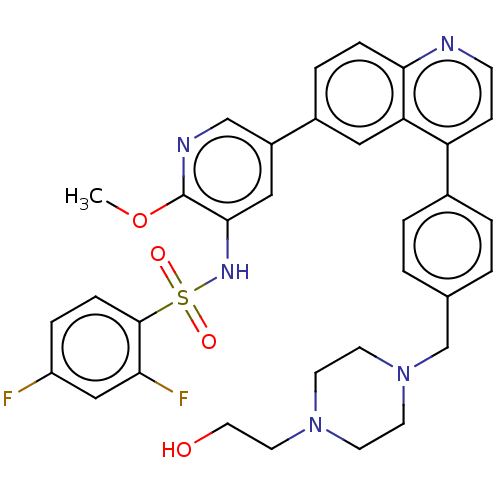
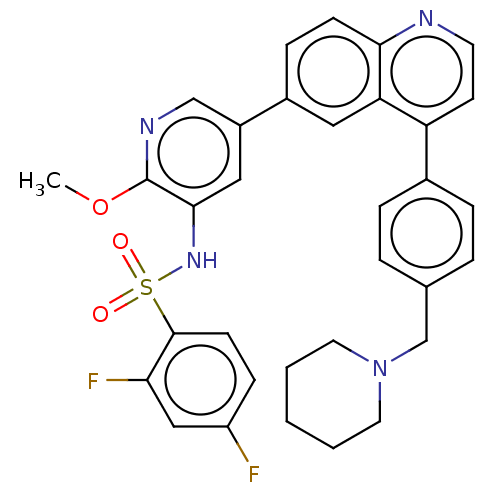
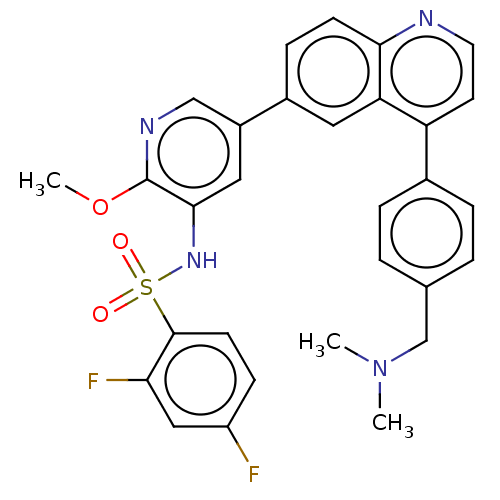
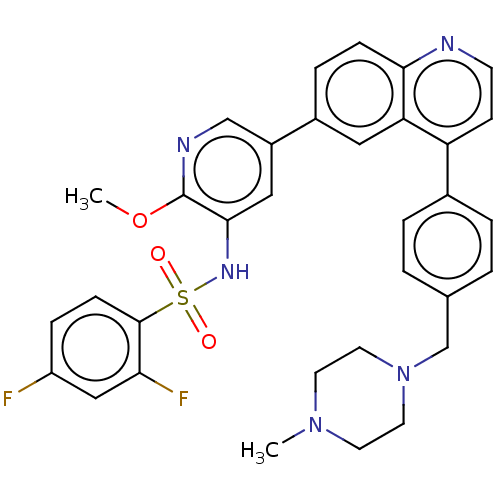
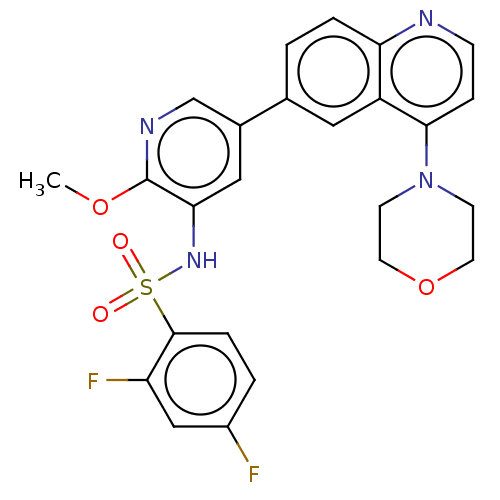
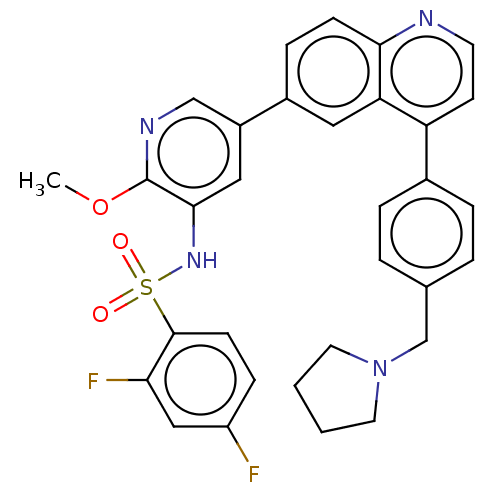
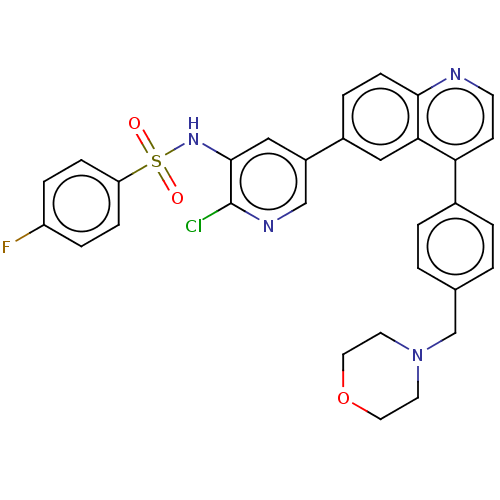
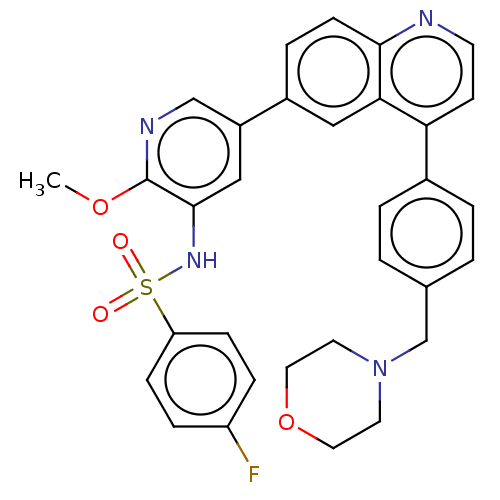
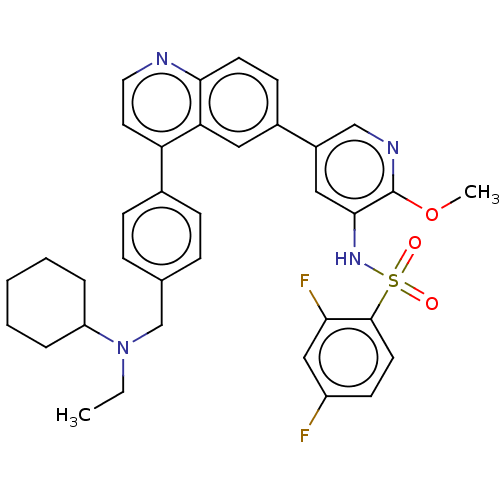
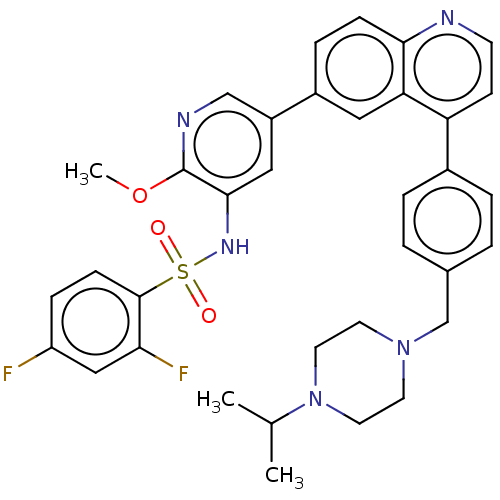
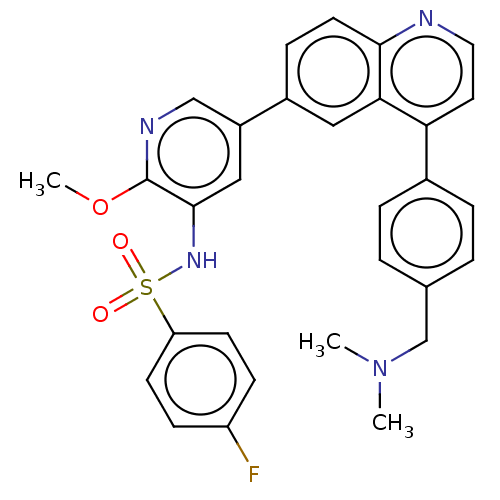
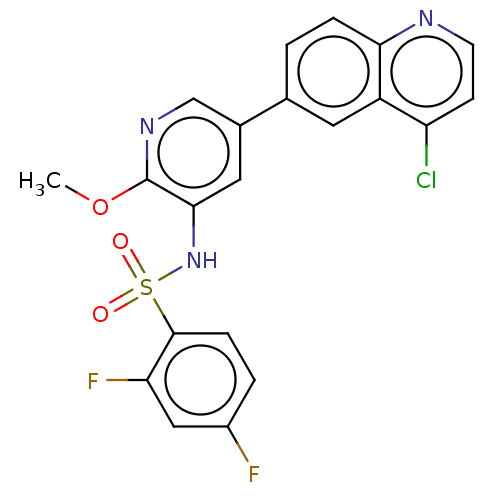
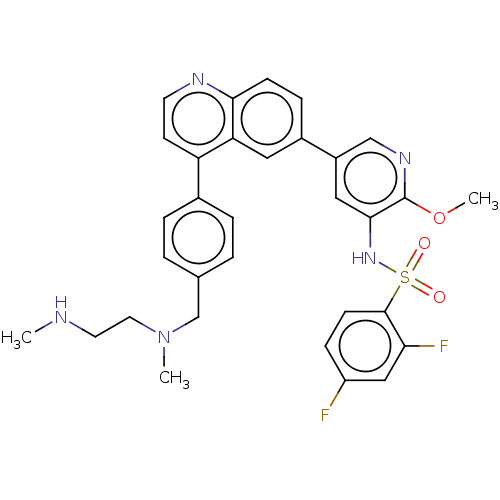
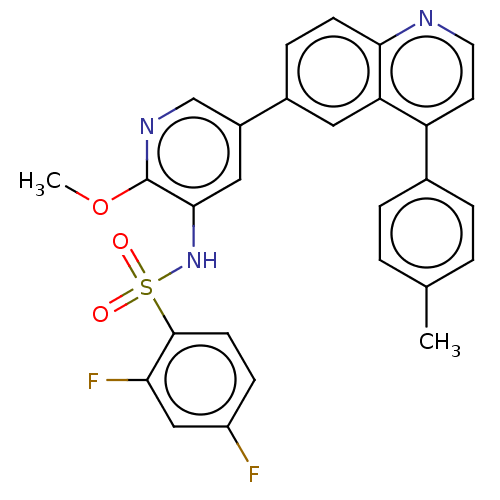
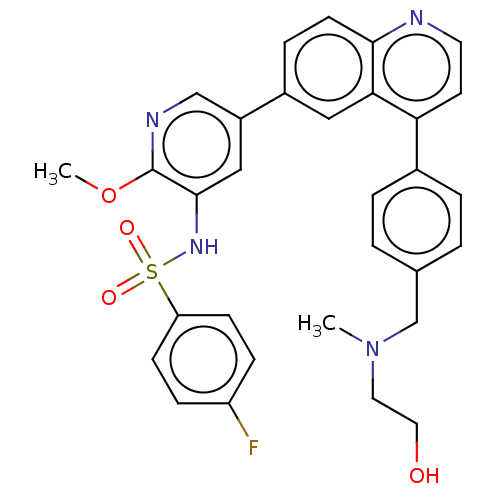
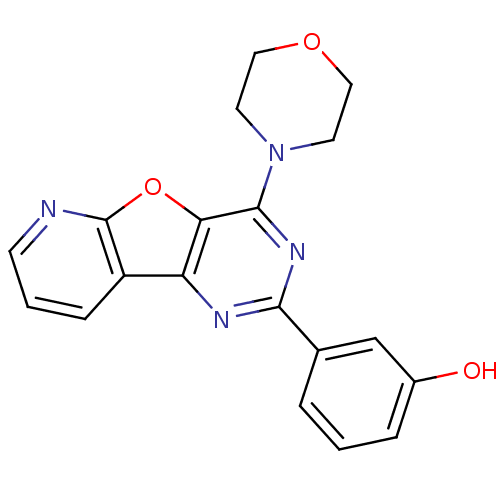
TargetSerine/threonine-protein kinase mTOR(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 0.310nMAssay Description:Inhibition of mTOR (unknown origin) using ULight-4E-BP1 peptide as substrate after 1 hr in presence of ATP by lance ultra assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 0.590nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 0.610nMAssay Description:Inhibition of PI3Kgamma (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by ADP-glo based luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 0.630nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 0.650nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 0.690nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 0.730nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 0.740nMAssay Description:Inhibition of PI3Kgamma (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by ADP-glo based luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 0.75nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 0.820nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 0.910nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 0.950nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetSerine/threonine-protein kinase mTOR(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.10nMAssay Description:Inhibition of mTOR (unknown origin) using ULight-4E-BP1 peptide as substrate after 1 hr in presence of ATP by lance ultra assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.10nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.10nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.10nMAssay Description:Inhibition of PI3Kbeta (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by ADP-glo based luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.10nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.10nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.10nMAssay Description:Inhibition of PI3Kgamma (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by ADP-glo based luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.10nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.20nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.20nMAssay Description:Inhibition of PI3Kbeta (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by ADP-glo based luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.20nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.30nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.30nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.30nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.40nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.40nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.5nMAssay Description:Inhibition of PI3Kgamma (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by ADP-glo based luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.5nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.60nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.60nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.70nMAssay Description:Inhibition of PI3Kbeta (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by ADP-glo based luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.70nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 1.80nMAssay Description:Inhibition of PI3Kbeta (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by ADP-glo based luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 2.10nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetSerine/threonine-protein kinase mTOR(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 2.20nMAssay Description:Inhibition of mTOR (unknown origin) using ULight-4E-BP1 peptide as substrate after 1 hr in presence of ATP by lance ultra assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 2.30nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 2.5nMAssay Description:Inhibition of PI3Kdelta (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 2.5nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 2.80nMAssay Description:Inhibition of PI3Kdelta (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 3.40nMAssay Description:Inhibition of PI3Kdelta (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetSerine/threonine-protein kinase mTOR(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 3.80nMAssay Description:Inhibition of mTOR (unknown origin) using ULight-4E-BP1 peptide as substrate after 1 hr in presence of ATP by lance ultra assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 3.80nMAssay Description:Inhibition of PI3Kdelta (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 4.5nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 4.80nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 6.10nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair
TargetSerine/threonine-protein kinase mTOR(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 8.80nMAssay Description:Inhibition of mTOR (unknown origin) using ULight-4E-BP1 peptide as substrate after 1 hr in presence of ATP by lance ultra assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Homo sapiens (Human))
Second Military Medical University
Curated by ChEMBL
Second Military Medical University
Curated by ChEMBL
Affinity DataIC50: 9.40nMAssay Description:Inhibition of PI3Kdelta (unknown origin) using PIP2 as substrate after 1 hr in presence of ATP by kinase-glo luminescence assayMore data for this Ligand-Target Pair

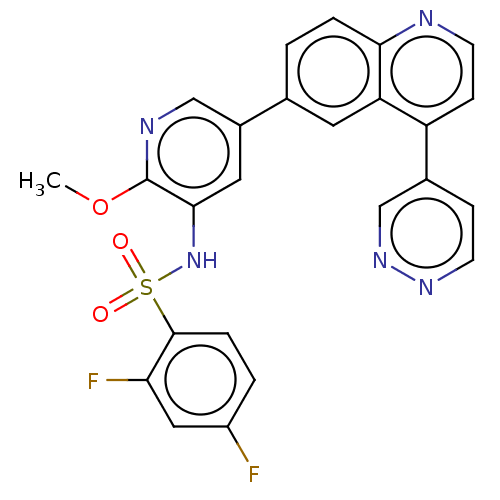
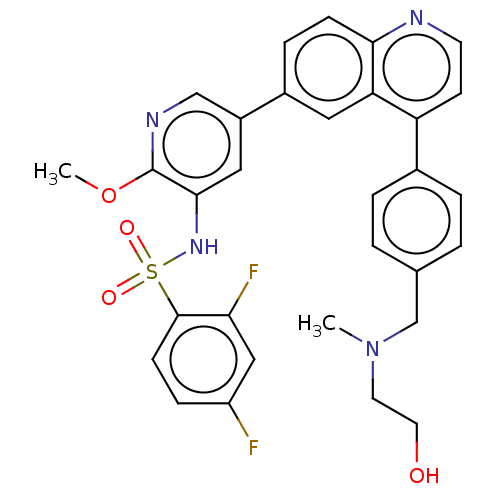
 3D Structure (crystal)
3D Structure (crystal)