Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Urokinase-type plasminogen activator
Ligand
BDBM16149
Substrate
BDBM14716
Meas. Tech.
Determination of Inhibitor Potency and Selectivity
Ki
66±n/a nM
Citation
 Fish, PV; Barber, CG; Brown, DG; Butt, R; Collis, MG; Dickinson, RP; Henry, BT; Horne, VA; Huggins, JP; King, E; O'gara, M; McCleverty, D; McIntosh, F; Phillips, C; Webster, R Selective Urokinase-Type Plasminogen Activator Inhibitors. 4. 1-(7-Sulfonamidoisoquinolinyl)guanidines. J Med Chem 50:2341-51 (2007) [PubMed] Article
Fish, PV; Barber, CG; Brown, DG; Butt, R; Collis, MG; Dickinson, RP; Henry, BT; Horne, VA; Huggins, JP; King, E; O'gara, M; McCleverty, D; McIntosh, F; Phillips, C; Webster, R Selective Urokinase-Type Plasminogen Activator Inhibitors. 4. 1-(7-Sulfonamidoisoquinolinyl)guanidines. J Med Chem 50:2341-51 (2007) [PubMed] Article More Info.:
Target
Name:
Urokinase-type plasminogen activator
Synonyms:
PLAU | U-plasminogen activator | UROK_HUMAN | Urokinase | Urokinase-type plasminogen activator (uPA) | Urokinase-type plasminogen activator/surface receptor
Type:
Enzyme
Mol. Mass.:
48528.62
Organism:
Homo sapiens (Human)
Description:
P00749
Residue:
431
Sequence:
MRALLARLLLCVLVVSDSKGSNELHQVPSNCDCLNGGTCVSNKYFSNIHWCNCPKKFGGQHCEIDKSKTCYEGNGHFYRGKASTDTMGRPCLPWNSATVLQQTYHAHRSDALQLGLGKHNYCRNPDNRRRPWCYVQVGLKPLVQECMVHDCADGKKPSSPPEELKFQCGQKTLRPRFKIIGGEFTTIENQPWFAAIYRRHRGGSVTYVCGGSLISPCWVISATHCFIDYPKKEDYIVYLGRSRLNSNTQGEMKFEVENLILHKDYSADTLAHHNDIALLKIRSKEGRCAQPSRTIQTICLPSMYNDPQFGTSCEITGFGKENSTDYLYPEQLKMTVVKLISHRECQQPHYYGSEVTTKMLCAADPQWKTDSCQGDSGGPLVCSLQGRMTLTGIVSWGRGCALKDKPGVYTRVSHFLPWIRSHTKEENGLAL
Inhibitor
Name:
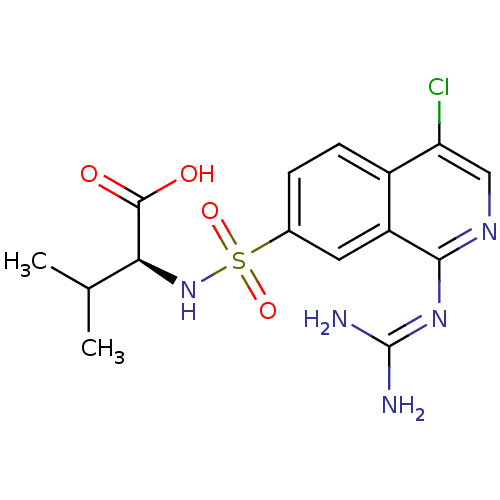
BDBM16149
Synonyms:
(2S)-2-({4-chloro-1-[(diaminomethylidene)amino]isoquinoline-7-}sulfonamido)-3-methylbutanoic acid | substituted glycine deriv. 23
Type:
Small organic molecule
Emp. Form.:
C15H18ClN5O4S
Mol. Mass.:
399.853
SMILES:
[#6]-[#6](-[#6])-[#6@H](-[#7]S(=O)(=O)c1ccc2c(Cl)cnc(\[#7]=[#6](/[#7])-[#7])c2c1)-[#6](-[#8])=O |r|
Substrate
Name:
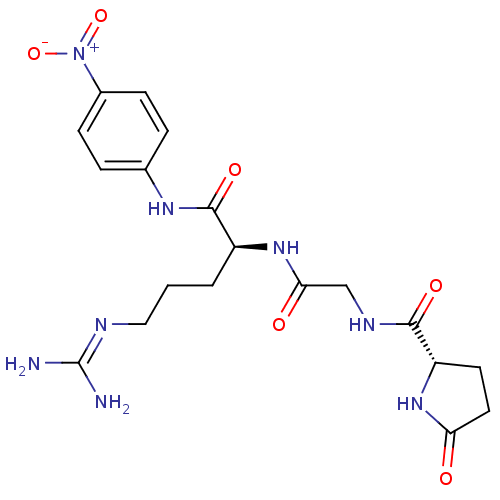
BDBM14716
Synonyms:
(2S)-5-carbamimidamido-N-(4-nitrophenyl)-2-(2-{[(2S)-5-oxopyrrolidin-2-yl]formamido}acetamido)pentanamide hydrochloride | Chromogenic Substrate S-2444 | L-Pyroglutamyl-glycyl-L-argininep-Nitroaniline hydrochloride
Type:
Small organic molecule
Emp. Form.:
C19H26N8O6
Mol. Mass.:
462.4597
SMILES:
[#7]\[#6](-[#7])=[#7]/[#6]-[#6]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6](=O)-[#7]-1)-[#6](=O)-[#7]-c1ccc(cc1)-[#7+](-[#8-])=O |r|