Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Carbonic anhydrase 5A, mitochondrial
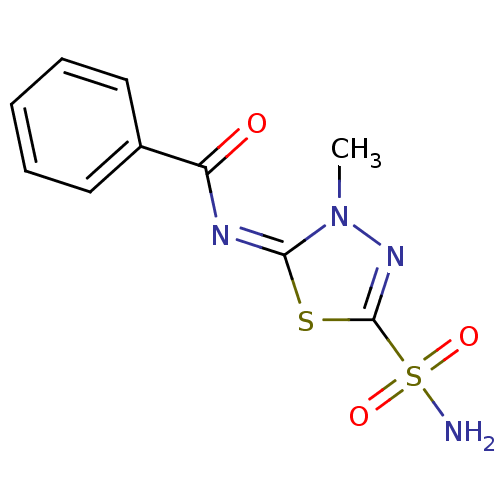
Ligand
BDBM16672
Substrate
BDBM10856
Meas. Tech.
Esterase Assay
Ki
83±n/a nM
Citation
 Vullo, D; Franchi, M; Gallori, E; Antel, J; Scozzafava, A; Supuran, CT Carbonic anhydrase inhibitors. Inhibition of mitochondrial isozyme V with aromatic and heterocyclic sulfonamides. J Med Chem 47:1272-9 (2004) [PubMed] Article
Vullo, D; Franchi, M; Gallori, E; Antel, J; Scozzafava, A; Supuran, CT Carbonic anhydrase inhibitors. Inhibition of mitochondrial isozyme V with aromatic and heterocyclic sulfonamides. J Med Chem 47:1272-9 (2004) [PubMed] Article More Info.:
Target
Name:
Carbonic anhydrase 5A, mitochondrial
Synonyms:
CAH5A_MOUSE | Ca5 | Ca5a | Car5 | Car5a | Carbonate dehydratase VA | Carbonic Anhydrase V | Carbonic Anhydrase VA | Carbonic anhydrase 5A, mitochondrial precursor
Type:
Enzyme
Mol. Mass.:
34081.17
Organism:
Mus musculus (mouse)
Description:
Full length murine, cloned isozyme.
Residue:
299
Sequence:
MLRRDPRKPLAILRHVGLLCATGPQRWRFQHSCAEEHSNCARHPLWTGPVSSAEGTRQSPINIQWKDSVYDPQLAPLRVSYDAASCRYLWNTGYFFQVEFDDSCEDSGISGGPLGNHYRLKQFHFHWGATDEWGSEHAVDGHTYPAELHLVHWNSTKYENYKKASVGENGLAVIGVFLKLGAHHQALQKLVDVLPEVRHKDTQVAMGPFDPSCLLPACRDYWTYPGSLTTPPLAESVTWIVQKTPVEVSPSQLSTFRTLLFSGRGEEEDVMVNNYRPLQPLRDRKLRSSFRLDRTKMRS
Inhibitor
Name:
BDBM16672
Synonyms:
5-Benzoylimino-4-methyl-delta2-1,3,4-thiadiazoline-2-sulfonamide | N-[(2E)-3-methyl-5-sulfamoyl-2,3-dihydro-1,3,4-thiadiazol-2-ylidene]benzamide | heterocyclic sulfonamide compound 37
Type:
Small organic molecule
Emp. Form.:
C10H10N4O3S2
Mol. Mass.:
298.341
SMILES:
Cn1nc(s\c1=N\C(=O)c1ccccc1)S(N)(=O)=O