Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Urotensin-2 receptor
Ligand
BDBM50164424
Substrate
n/a
Meas. Tech.
ChEMBL_342488 (CHEMBL861584)
EC50
1122±n/a nM
Citation
 Lehmann, F; Pettersen, A; Currier, EA; Sherbukhin, V; Olsson, R; Hacksell, U; Luthman, K Novel potent and efficacious nonpeptidic urotensin II receptor agonists. J Med Chem 49:2232-40 (2006) [PubMed] Article
Lehmann, F; Pettersen, A; Currier, EA; Sherbukhin, V; Olsson, R; Hacksell, U; Luthman, K Novel potent and efficacious nonpeptidic urotensin II receptor agonists. J Med Chem 49:2232-40 (2006) [PubMed] Article More Info.:
Target
Name:
Urotensin-2 receptor
Synonyms:
G-protein coupled receptor 14 | GPR14 | UR-II-R | UR2R_HUMAN | UTS2R | Urotensin II receptor | Urotensin-II
Type:
Enzyme Catalytic Domain
Mol. Mass.:
42159.71
Organism:
Homo sapiens (Human)
Description:
Urotensin-II UTS2R HUMAN::Q9UKP6
Residue:
389
Sequence:
MALTPESPSSFPGLAATGSSVPEPPGGPNATLNSSWASPTEPSSLEDLVATGTIGTLLSAMGVVGVVGNAYTLVVTCRSLRAVASMYVYVVNLALADLLYLLSIPFIVATYVTKEWHFGDVGCRVLFGLDFLTMHASIFTLTVMSSERYAAVLRPLDTVQRPKGYRKLLALGTWLLALLLTLPVMLAMRLVRRGPKSLCLPAWGPRAHRAYLTLLFATSIAGPGLLIGLLYARLARAYRRSQRASFKRARRPGARALRLVLGIVLLFWACFLPFWLWQLLAQYHQAPLAPRTARIVNYLTTCLTYGNSCANPFLYTLLTRNYRDHLRGRVRGPGSGGGRGPVPSLQPRARFQRCSGRSLSSCSPQPTDSLVLAPAAPARPAPEGPRAPA
Inhibitor
Name:
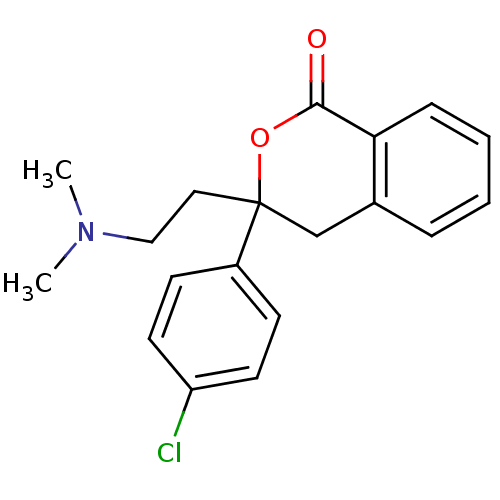
BDBM50164424
Synonyms:
3-(4-Chloro-phenyl)-3-(2-dimethylamino-ethyl)-isochroman-1-one | 3-(4-chlorophenyl)-3-(2-(dimethylamino)ethyl)isochroman-1-one | CHEMBL192359
Type:
Small organic molecule
Emp. Form.:
C19H20ClNO2
Mol. Mass.:
329.821
SMILES:
CN(C)CCC1(Cc2ccccc2C(=O)O1)c1ccc(Cl)cc1