Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Genome polyprotein
Ligand
BDBM50112651
Substrate
n/a
Meas. Tech.
ChEMBL_161092 (CHEMBL771553)
EC50
160±n/a nM
Citation
 Johnson, TO; Hua, Y; Luu, HT; Brown, EL; Chan, F; Chu, SS; Dragovich, PS; Eastman, BW; Ferre, RA; Fuhrman, SA; Hendrickson, TF; Maldonado, FC; Matthews, DA; Meador, JW; Patick, AK; Reich, SH; Skalitzky, DJ; Worland, ST; Yang, M; Zalman, LS Structure-based design of a parallel synthetic array directed toward the discovery of irreversible inhibitors of human rhinovirus 3C protease. J Med Chem 45:2016-23 (2002) [PubMed] Article
Johnson, TO; Hua, Y; Luu, HT; Brown, EL; Chan, F; Chu, SS; Dragovich, PS; Eastman, BW; Ferre, RA; Fuhrman, SA; Hendrickson, TF; Maldonado, FC; Matthews, DA; Meador, JW; Patick, AK; Reich, SH; Skalitzky, DJ; Worland, ST; Yang, M; Zalman, LS Structure-based design of a parallel synthetic array directed toward the discovery of irreversible inhibitors of human rhinovirus 3C protease. J Med Chem 45:2016-23 (2002) [PubMed] Article More Info.:
Target
Name:
Genome polyprotein
Synonyms:
Human rhinovirus A protease | Human rhinovirus B 3A protease
Type:
PROTEIN
Mol. Mass.:
44361.04
Organism:
Human rhinovirus B
Description:
ChEMBL_158953
Residue:
401
Sequence:
AFRPCNVNTKIGNAKCCPFVCGKAVTFKDRSTCSTYNLSSSLHHILEEDKRRRQVVDVMSAIFQGPISLDAPPPPAIADLLQSVRTPRVIKYCQIIMGHPAECQVERDLNIANSIIAIIANIISIAGIIFVIYKLFCSLQGPYSGEPKPKTKVPERRVVAQGPEEEFGRSILKNNTCVITTGNGKFTGLGIHDRILIIPTHADPGREVQVNGVHTKVLDSYDLYNRDGVKLEITVIQLDRNEKFRDIRKYIPETEDDYPECNLALSANQDEPTIIKVGDVVSYGNILLSGNQTARMLKYNYPTKSGYCGGVLYKIGQILGIHVGGNGRDGFSAMLLRSYFTGQIKVNKHATECGLPDIQTIHTPSKTKLQPSVFYDVFPGSKEPAVLTDNDPRLEVNFKEA
Inhibitor
Name:
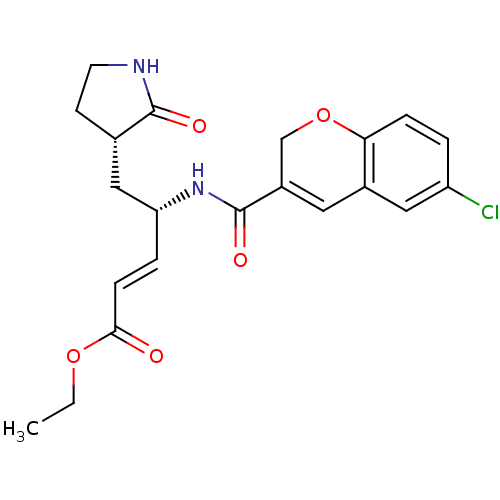
BDBM50112651
Synonyms:
4-[(6-Chloro-2H-chromene-3-carbonyl)-amino]-5-(2-oxo-pyrrolidin-3-yl)-pent-2-enoic acid ethyl ester | 4S-[(6-Chloro-2H-chromene-3-carbonyl)amino]-5-(2-oxopyrrolidin-3S-yl)pent-2-enoic Acid Ethyl Ester | CHEMBL293441
Type:
Small organic molecule
Emp. Form.:
C21H23ClN2O5
Mol. Mass.:
418.871
SMILES:
CCOC(=O)\C=C\[C@H](C[C@@H]1CCNC1=O)NC(=O)C1=Cc2cc(Cl)ccc2OC1 |r,t:19|