Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
C-C chemokine receptor type 5
Ligand
BDBM50145681
Substrate
n/a
Meas. Tech.
ChEMBL_39636 (CHEMBL649943)
Ki
1.4±n/a nM
Citation
 Tagat, JR; McCombie, SW; Nazareno, D; Labroli, MA; Xiao, Y; Steensma, RW; Strizki, JM; Baroudy, BM; Cox, K; Lachowicz, J; Varty, G; Watkins, R Piperazine-based CCR5 antagonists as HIV-1 inhibitors. IV. Discovery of 1-[(4,6-dimethyl-5-pyrimidinyl)carbonyl]- 4-[4-[2-methoxy-1(R)-4-(trifluoromethyl)phenyl]ethyl-3(S)-methyl-1-piperazinyl]- 4-methylpiperidine (Sch-417690/Sch-D), a potent, highly selective, and orally bioavailable CCR5 antagoni J Med Chem 47:2405-8 (2004) [PubMed] Article
Tagat, JR; McCombie, SW; Nazareno, D; Labroli, MA; Xiao, Y; Steensma, RW; Strizki, JM; Baroudy, BM; Cox, K; Lachowicz, J; Varty, G; Watkins, R Piperazine-based CCR5 antagonists as HIV-1 inhibitors. IV. Discovery of 1-[(4,6-dimethyl-5-pyrimidinyl)carbonyl]- 4-[4-[2-methoxy-1(R)-4-(trifluoromethyl)phenyl]ethyl-3(S)-methyl-1-piperazinyl]- 4-methylpiperidine (Sch-417690/Sch-D), a potent, highly selective, and orally bioavailable CCR5 antagoni J Med Chem 47:2405-8 (2004) [PubMed] Article More Info.:
Target
Name:
C-C chemokine receptor type 5
Synonyms:
C-C CKR-5 | C-C chemokine receptor type 5 | CC-CKR-5 | CCR-5 | CCR5 | CCR5/mu opioid receptor complex | CCR5_HUMAN | CD_antigen=CD195 | CHEMR13 | CMKBR5 | HIV-1 fusion coreceptor
Type:
Enzyme
Mol. Mass.:
40540.21
Organism:
Homo sapiens (Human)
Description:
P51681
Residue:
352
Sequence:
MDYQVSSPIYDINYYTSEPCQKINVKQIAARLLPPLYSLVFIFGFVGNMLVILILINCKRLKSMTDIYLLNLAISDLFFLLTVPFWAHYAAAQWDFGNTMCQLLTGLYFIGFFSGIFFIILLTIDRYLAVVHAVFALKARTVTFGVVTSVITWVVAVFASLPGIIFTRSQKEGLHYTCSSHFPYSQYQFWKNFQTLKIVILGLVLPLLVMVICYSGILKTLLRCRNEKKRHRAVRLIFTIMIVYFLFWAPYNIVLLLNTFQEFFGLNNCSSSNRLDQAMQVTETLGMTHCCINPIIYAFVGEKFRNYLLVFFQKHIAKRFCKCCSIFQQEAPERASSVYTRSTGEQEISVGL
Inhibitor
Name:
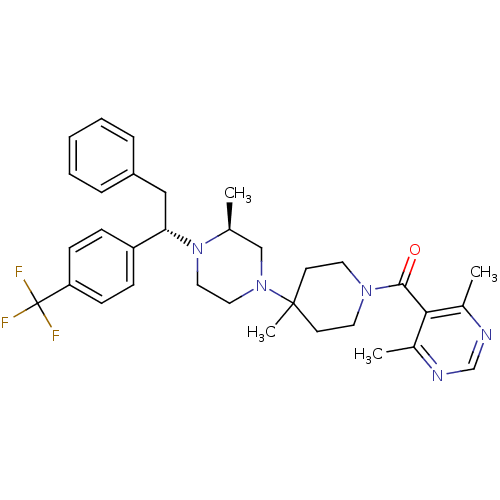
BDBM50145681
Synonyms:
(4,6-Dimethyl-pyrimidin-5-yl)-(4-methyl-4-{(S)-3-methyl-4-[(S)-2-phenyl-1-(4-trifluoromethyl-phenyl)-ethyl]-piperazin-1-yl}-piperidin-1-yl)-methanone | CHEMBL79061
Type:
Small organic molecule
Emp. Form.:
C33H40F3N5O
Mol. Mass.:
579.6988
SMILES:
C[C@H]1CN(CCN1[C@@H](Cc1ccccc1)c1ccc(cc1)C(F)(F)F)C1(C)CCN(CC1)C(=O)c1c(C)ncnc1C