Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
RAC-alpha serine/threonine-protein kinase
Ligand
BDBM50188913
Substrate
n/a
Meas. Tech.
ChEMBL_389600 (CHEMBL868045)
IC50
6±n/a nM
Citation
 Lin, X; Murray, JM; Rico, AC; Wang, MX; Chu, DT; Zhou, Y; Del Rosario, M; Kaufman, S; Ma, S; Fang, E; Crawford, K; Jefferson, AB Discovery of 2-pyrimidyl-5-amidothiophenes as potent inhibitors for AKT: synthesis and SAR studies. Bioorg Med Chem Lett 16:4163-8 (2006) [PubMed] Article
Lin, X; Murray, JM; Rico, AC; Wang, MX; Chu, DT; Zhou, Y; Del Rosario, M; Kaufman, S; Ma, S; Fang, E; Crawford, K; Jefferson, AB Discovery of 2-pyrimidyl-5-amidothiophenes as potent inhibitors for AKT: synthesis and SAR studies. Bioorg Med Chem Lett 16:4163-8 (2006) [PubMed] Article More Info.:
Target
Name:
RAC-alpha serine/threonine-protein kinase
Synonyms:
AKT phosphorylation (p-AKT) | AKT1 | AKT1/PPP1CA | AKT1_HUMAN | C-AKT | PKB | PKB alpha | Protein kinase Akt-1 | Protein kinase B | Protein kinase B (AKT1) | Protein kinase B (Akt 1) | Protein kinase B (Akt) | Protein kinase B alpha | Protein kinase B alpha (AKT1) | Proto-oncogene Akt (Akt1) | Proto-oncogene c-Akt (AKT) | Proto-oncogene c-Akt (AKT1) | RAC | RAC-PK-alpha | RAC-alpha serine/threonine-protein kinase (AKT) | RAC-alpha serine/threonine-protein kinase (AKT1) | RAC-alpha serine/threonine-protein kinase (pAKT)
Type:
Enzyme
Mol. Mass.:
55681.25
Organism:
Homo sapiens (Human)
Description:
P31749
Residue:
480
Sequence:
MSDVAIVKEGWLHKRGEYIKTWRPRYFLLKNDGTFIGYKERPQDVDQREAPLNNFSVAQCQLMKTERPRPNTFIIRCLQWTTVIERTFHVETPEEREEWTTAIQTVADGLKKQEEEEMDFRSGSPSDNSGAEEMEVSLAKPKHRVTMNEFEYLKLLGKGTFGKVILVKEKATGRYYAMKILKKEVIVAKDEVAHTLTENRVLQNSRHPFLTALKYSFQTHDRLCFVMEYANGGELFFHLSRERVFSEDRARFYGAEIVSALDYLHSEKNVVYRDLKLENLMLDKDGHIKITDFGLCKEGIKDGATMKTFCGTPEYLAPEVLEDNDYGRAVDWWGLGVVMYEMMCGRLPFYNQDHEKLFELILMEEIRFPRTLGPEAKSLLSGLLKKDPKQRLGGGSEDAKEIMQHRFFAGIVWQHVYEKKLSPPFKPQVTSETDTRYFDEEFTAQMITITPPDQDDSMECVDSERRPHFPQFSYSASGTA
Inhibitor
Name:
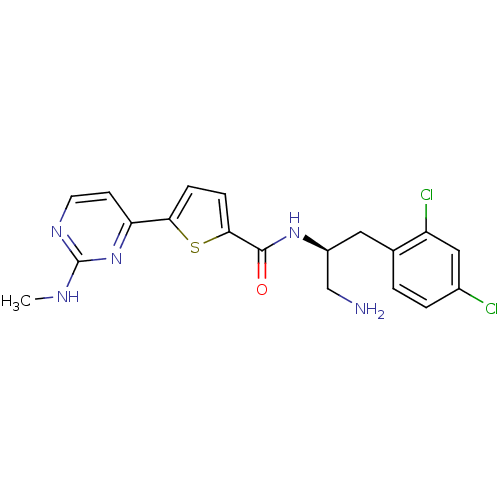
BDBM50188913
Synonyms:
CHEMBL213618 | N-((S)-1-amino-3-(2,4-dichlorophenyl)propan-2-yl)-5-(2-(methylamino)pyrimidin-4-yl)thiophene-2-carboxamide | N-[(1S)-2-AMINO-1-(2,4-DICHLOROBENZYL)ETHYL]-5-[2-(METHYLAMINO)PYRIMIDIN-4-YL]THIOPHENE-2-CARBOXAMIDE
Type:
Small organic molecule
Emp. Form.:
C19H19Cl2N5OS
Mol. Mass.:
436.358
SMILES:
CNc1nccc(n1)-c1ccc(s1)C(=O)N[C@H](CN)Cc1ccc(Cl)cc1Cl