Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Bifunctional epoxide hydrolase 2
Ligand
BDBM25738
Substrate
n/a
Meas. Tech.
ChEMBL_446700 (CHEMBL896996)
IC50
14±n/a nM
Citation
 Kim, IH; Tsai, HJ; Nishi, K; Kasagami, T; Morisseau, C; Hammock, BD 1,3-disubstituted ureas functionalized with ether groups are potent inhibitors of the soluble epoxide hydrolase with improved pharmacokinetic properties. J Med Chem 50:5217-26 (2007) [PubMed] Article
Kim, IH; Tsai, HJ; Nishi, K; Kasagami, T; Morisseau, C; Hammock, BD 1,3-disubstituted ureas functionalized with ether groups are potent inhibitors of the soluble epoxide hydrolase with improved pharmacokinetic properties. J Med Chem 50:5217-26 (2007) [PubMed] Article More Info.:
Target
Name:
Bifunctional epoxide hydrolase 2
Synonyms:
Cytosolic epoxide hydrolase 2 | EBifunctional epoxide hydrolase 2 | EPHX2 | Epoxide hydratase | HYES_HUMAN | Lipid-phosphate phosphatase | Soluble epoxide hydrolase (sEH) | epoxide hydrolase 2, cytoplasmic
Type:
Enzyme
Mol. Mass.:
62613.07
Organism:
Homo sapiens (Human)
Description:
P34913
Residue:
555
Sequence:
MTLRAAVFDLDGVLALPAVFGVLGRTEEALALPRGLLNDAFQKGGPEGATTRLMKGEITLSQWIPLMEENCRKCSETAKVCLPKNFSIKEIFDKAISARKINRPMLQAALMLRKKGFTTAILTNTWLDDRAERDGLAQLMCELKMHFDFLIESCQVGMVKPEPQIYKFLLDTLKASPSEVVFLDDIGANLKPARDLGMVTILVQDTDTALKELEKVTGIQLLNTPAPLPTSCNPSDMSHGYVTVKPRVRLHFVELGSGPAVCLCHGFPESWYSWRYQIPALAQAGYRVLAMDMKGYGESSAPPEIEEYCMEVLCKEMVTFLDKLGLSQAVFIGHDWGGMLVWYMALFYPERVRAVASLNTPFIPANPNMSPLESIKANPVFDYQLYFQEPGVAEAELEQNLSRTFKSLFRASDESVLSMHKVCEAGGLFVNSPEEPSLSRMVTEEEIQFYVQQFKKSGFRGPLNWYRNMERNWKWACKSLGRKILIPALMVTAEKDFVLVPQMSQHMEDWIPHLKRGHIEDCGHWTQMDKPTEVNQILIKWLDSDARNPPVVSKM
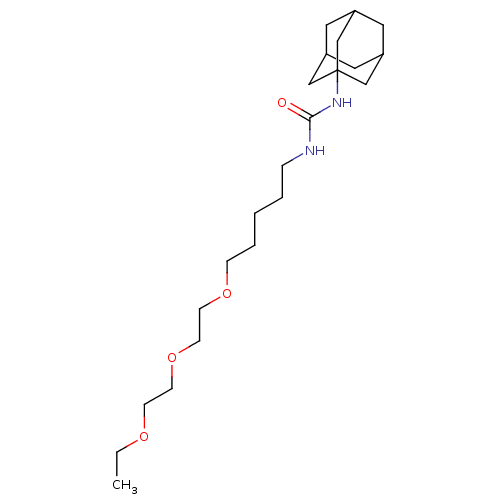
Inhibitor
Name:
BDBM25738
Synonyms:
1-adamantan-1-yl-3-{5-[2-(2-ethoxyethoxy)ethoxy]pentyl}urea | CHEMBL242655 | US10383835, Compound 950 | Urea-based compound, 19
Type:
Small organic molecule
Emp. Form.:
C22H40N2O4
Mol. Mass.:
396.564
SMILES:
CCOCCOCCOCCCCCNC(=O)NC12CC3CC(CC(C3)C1)C2 |TLB:25:20:27:24.23.26,25:24:20.21.19:27,THB:23:22:19:24.25.26,23:24:19:22.21.27|