Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Muscarinic acetylcholine receptor M3
Ligand
BDBM43822
Substrate
n/a
Meas. Tech.
ChEMBL_788149 (CHEMBL1919230)
EC50
>30000±n/a nM
Citation
 Lebois, EP; Digby, GJ; Sheffler, DJ; Melancon, BJ; Tarr, JC; Cho, HP; Miller, NR; Morrison, R; Bridges, TM; Xiang, Z; Scott Daniels, J; Wood, MR; Conn, PJ; Lindsley, CW Development of a highly selective, orally bioavailable and CNS penetrant M1 agonist derived from the MLPCN probe ML071. Bioorg Med Chem Lett 21:6451-5 (2011) [PubMed] Article
Lebois, EP; Digby, GJ; Sheffler, DJ; Melancon, BJ; Tarr, JC; Cho, HP; Miller, NR; Morrison, R; Bridges, TM; Xiang, Z; Scott Daniels, J; Wood, MR; Conn, PJ; Lindsley, CW Development of a highly selective, orally bioavailable and CNS penetrant M1 agonist derived from the MLPCN probe ML071. Bioorg Med Chem Lett 21:6451-5 (2011) [PubMed] Article More Info.:
Target
Name:
Muscarinic acetylcholine receptor M3
Synonyms:
ACM3_RAT | Cholinergic, muscarinic M3 | Chrm-3 | Chrm3
Type:
Enzyme Catalytic Domain
Mol. Mass.:
66086.66
Organism:
RAT
Description:
Cholinergic, muscarinic M3 CHRM3 RAT::P08483
Residue:
589
Sequence:
MTLHSNSTTSPLFPNISSSWVHSPSEAGLPLGTVTQLGSYNISQETGNFSSNDTSSDPLGGHTIWQVVFIAFLTGFLALVTIIGNILVIVAFKVNKQLKTVNNYFLLSLACADLIIGVISMNLFTTYIIMNRWALGNLACDLWLSIDYVASNASVMNLLVISFDRYFSITRPLTYRAKRTTKRAGVMIGLAWVISFVLWAPAILFWQYFVGKRTVPPGECFIQFLSEPTITFGTAIAAFYMPVTIMTILYWRIYKETEKRTKELAGLQASGTEAEAENFVHPTGSSRSCSSYELQQQGVKRSSRRKYGRCHFWFTTKSWKPSAEQMDQDHSSSDSWNNNDAAASLENSASSDEEDIGSETRAIYSIVLKLPGHSSILNSTKLPSSDNLQVSNEDLGTVDVERNAHKLQAQKSMGDGDNCQKDFTKLPIQLESAVDTGKTSDTNSSADKTTATLPLSFKEATLAKRFALKTRSQITKRKRMSLIKEKKAAQTLSAILLAFIITWTPYNIMVLVNTFCDSCIPKTYWNLGYWLCYINSTVNPVCYALCNKTFRTTFKTLLLCQCDKRKRRKQQYQQRQSVIFHKRVPEQAL
Inhibitor
Name:
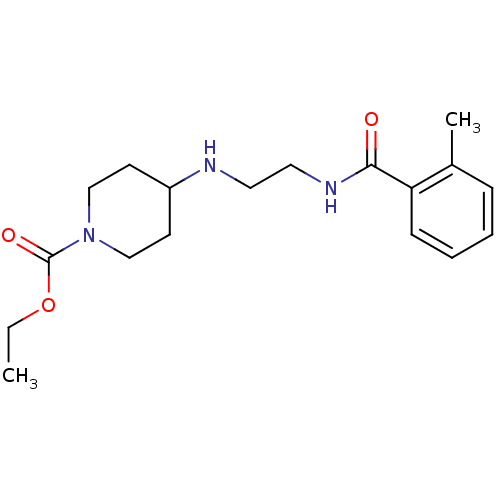
BDBM43822
Synonyms:
4-[2-(o-toluoylamino)ethylamino]piperidine-1-carboxylic acid ethyl ester;hydrochloride | 4-[2-[[(2-methylphenyl)-oxomethyl]amino]ethylamino]-1-piperidinecarboxylic acid ethyl ester;hydrochloride | CHEMBL1624037 | VU0357017 | cid_25010775 | ethyl 4-[2-[(2-methylbenzoyl)amino]ethylamino]piperidine-1-carboxylate;hydrochloride | ethyl 4-[2-[(2-methylphenyl)carbonylamino]ethylamino]piperidine-1-carboxylate;hydrochloride
Type:
Small organic molecule
Emp. Form.:
C18H27N3O3
Mol. Mass.:
333.4253
SMILES:
CCOC(=O)N1CCC(CC1)NCCNC(=O)c1ccccc1C