Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Adenosine receptor A2a
Ligand
BDBM50163027
Substrate
n/a
Meas. Tech.
ChEMBL_479906 (CHEMBL935759)
Ki
10000±n/a nM
Citation
 Melman, A; Gao, ZG; Kumar, D; Wan, TC; Gizewski, E; Auchampach, JA; Jacobson, KA Design of (N)-methanocarba adenosine 5'-uronamides as species-independent A3 receptor-selective agonists. Bioorg Med Chem Lett 18:2813-9 (2008) [PubMed] Article
Melman, A; Gao, ZG; Kumar, D; Wan, TC; Gizewski, E; Auchampach, JA; Jacobson, KA Design of (N)-methanocarba adenosine 5'-uronamides as species-independent A3 receptor-selective agonists. Bioorg Med Chem Lett 18:2813-9 (2008) [PubMed] Article More Info.:
Target
Name:
Adenosine receptor A2a
Synonyms:
A2A adenosine receptor (hA2A) | AA2AR_HUMAN | ADENOSINE A2 | ADENOSINE A2a | ADORA2 | ADORA2A | Adenosine A2A receptor (A2AAR)
Type:
G Protein-Coupled Receptor (GPCR)
Mol. Mass.:
44716.46
Organism:
Homo sapiens (Human)
Description:
P29274
Residue:
412
Sequence:
MPIMGSSVYITVELAIAVLAILGNVLVCWAVWLNSNLQNVTNYFVVSLAAADIAVGVLAIPFAITISTGFCAACHGCLFIACFVLVLTQSSIFSLLAIAIDRYIAIRIPLRYNGLVTGTRAKGIIAICWVLSFAIGLTPMLGWNNCGQPKEGKNHSQGCGEGQVACLFEDVVPMNYMVYFNFFACVLVPLLLMLGVYLRIFLAARRQLKQMESQPLPGERARSTLQKEVHAAKSLAIIVGLFALCWLPLHIINCFTFFCPDCSHAPLWLMYLAIVLSHTNSVVNPFIYAYRIREFRQTFRKIIRSHVLRQQEPFKAAGTSARVLAAHGSDGEQVSLRLNGHPPGVWANGSAPHPERRPNGYALGLVSGGSAQESQGNTGLPDVELLSHELKGVCPEPPGLDDPLAQDGAGVS
Inhibitor
Name:
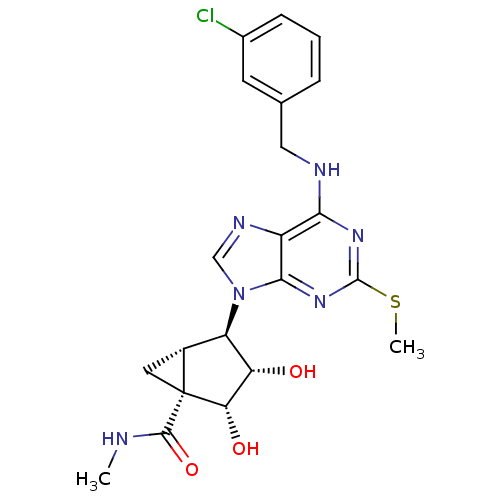
BDBM50163027
Synonyms:
(1S,2R,3S,4R,5S)-4-(6-(3-chlorobenzylamino)-2-(methylthio)-9H-purin-9-yl)-2,3-dihydroxy-N-methylbicyclo[3.1.0]hexane-1-carboxamide | 1N-methyl-4-[6-(3-chlorobenzylamino)-2-methylsulfanyl-9H-9-purinyl]-2,3-dihydroxy-(1S,2R,3S,4R,5S)-bicyclo[3.1.0]hexane-1-carboxamide | CHEMBL175814
Type:
Small organic molecule
Emp. Form.:
C21H23ClN6O3S
Mol. Mass.:
474.964
SMILES:
CNC(=O)[C@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NCc3cccc(Cl)c3)nc(SC)nc12