Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Insulin receptor
Ligand
BDBM50193995
Substrate
n/a
Meas. Tech.
ChEMBL_542081 (CHEMBL1016950)
IC50
>10000±n/a nM
Citation
 Changelian, PS; Flanagan, ME; Ball, DJ; Kent, CR; Magnuson, KS; Martin, WH; Rizzuti, BJ; Sawyer, PS; Perry, BD; Brissette, WH; McCurdy, SP; Kudlacz, EM; Conklyn, MJ; Elliott, EA; Koslov, ER; Fisher, MB; Strelevitz, TJ; Yoon, K; Whipple, DA; Sun, J; Munchhof, MJ; Doty, JL; Casavant, JM; Blumenkopf, TA; Hines, M; Brown, MF; Lillie, BM; Subramanyam, C; Shang-Poa, C; Milici, AJ; Beckius, GE; Moyer, JD; Su, C; Woodworth, TG; Gaweco, AS; Beals, CR; Littman, BH; Fisher, DA; Smith, JF; Zagouras, P; Magna, HA; Saltarelli, MJ; Johnson, KS; Nelms, LF; Des Etages, SG; Hayes, LS; Kawabata, TT; Finco-Kent, D; Baker, DL; Larson, M; Si, MS; Paniagua, R; Higgins, J; Holm, B; Reitz, B; Zhou, YJ; Morris, RE; O'Shea, JJ; Borie, DC Prevention of organ allograft rejection by a specific Janus kinase 3 inhibitor. Science 302:875-878 (2003) [PubMed] Article
Changelian, PS; Flanagan, ME; Ball, DJ; Kent, CR; Magnuson, KS; Martin, WH; Rizzuti, BJ; Sawyer, PS; Perry, BD; Brissette, WH; McCurdy, SP; Kudlacz, EM; Conklyn, MJ; Elliott, EA; Koslov, ER; Fisher, MB; Strelevitz, TJ; Yoon, K; Whipple, DA; Sun, J; Munchhof, MJ; Doty, JL; Casavant, JM; Blumenkopf, TA; Hines, M; Brown, MF; Lillie, BM; Subramanyam, C; Shang-Poa, C; Milici, AJ; Beckius, GE; Moyer, JD; Su, C; Woodworth, TG; Gaweco, AS; Beals, CR; Littman, BH; Fisher, DA; Smith, JF; Zagouras, P; Magna, HA; Saltarelli, MJ; Johnson, KS; Nelms, LF; Des Etages, SG; Hayes, LS; Kawabata, TT; Finco-Kent, D; Baker, DL; Larson, M; Si, MS; Paniagua, R; Higgins, J; Holm, B; Reitz, B; Zhou, YJ; Morris, RE; O'Shea, JJ; Borie, DC Prevention of organ allograft rejection by a specific Janus kinase 3 inhibitor. Science 302:875-878 (2003) [PubMed] Article More Info.:
Target
Name:
Insulin receptor
Synonyms:
INSR | INSR protein | INSR_HUMAN | Insulin receptor (IR) | Insulin receptor beta subunit
Type:
Receptor Tyrosine Kinase
Mol. Mass.:
156322.60
Organism:
Homo sapiens (Human)
Description:
P06213
Residue:
1382
Sequence:
MATGGRRGAAAAPLLVAVAALLLGAAGHLYPGEVCPGMDIRNNLTRLHELENCSVIEGHLQILLMFKTRPEDFRDLSFPKLIMITDYLLLFRVYGLESLKDLFPNLTVIRGSRLFFNYALVIFEMVHLKELGLYNLMNITRGSVRIEKNNELCYLATIDWSRILDSVEDNYIVLNKDDNEECGDICPGTAKGKTNCPATVINGQFVERCWTHSHCQKVCPTICKSHGCTAEGLCCHSECLGNCSQPDDPTKCVACRNFYLDGRCVETCPPPYYHFQDWRCVNFSFCQDLHHKCKNSRRQGCHQYVIHNNKCIPECPSGYTMNSSNLLCTPCLGPCPKVCHLLEGEKTIDSVTSAQELRGCTVINGSLIINIRGGNNLAAELEANLGLIEEISGYLKIRRSYALVSLSFFRKLRLIRGETLEIGNYSFYALDNQNLRQLWDWSKHNLTITQGKLFFHYNPKLCLSEIHKMEEVSGTKGRQERNDIALKTNGDQASCENELLKFSYIRTSFDKILLRWEPYWPPDFRDLLGFMLFYKEAPYQNVTEFDGQDACGSNSWTVVDIDPPLRSNDPKSQNHPGWLMRGLKPWTQYAIFVKTLVTFSDERRTYGAKSDIIYVQTDATNPSVPLDPISVSNSSSQIILKWKPPSDPNGNITHYLVFWERQAEDSELFELDYCLKGLKLPSRTWSPPFESEDSQKHNQSEYEDSAGECCSCPKTDSQILKELEESSFRKTFEDYLHNVVFVPRKTSSGTGAEDPRPSRKRRSLGDVGNVTVAVPTVAAFPNTSSTSVPTSPEEHRPFEKVVNKESLVISGLRHFTGYRIELQACNQDTPEERCSVAAYVSARTMPEAKADDIVGPVTHEIFENNVVHLMWQEPKEPNGLIVLYEVSYRRYGDEELHLCVSRKHFALERGCRLRGLSPGNYSVRIRATSLAGNGSWTEPTYFYVTDYLDVPSNIAKIIIGPLIFVFLFSVVIGSIYLFLRKRQPDGPLGPLYASSNPEYLSASDVFPCSVYVPDEWEVSREKITLLRELGQGSFGMVYEGNARDIIKGEAETRVAVKTVNESASLRERIEFLNEASVMKGFTCHHVVRLLGVVSKGQPTLVVMELMAHGDLKSYLRSLRPEAENNPGRPPPTLQEMIQMAAEIADGMAYLNAKKFVHRDLAARNCMVAHDFTVKIGDFGMTRDIYETDYYRKGGKGLLPVRWMAPESLKDGVFTTSSDMWSFGVVLWEITSLAEQPYQGLSNEQVLKFVMDGGYLDQPDNCPERVTDLMRMCWQFNPKMRPTFLEIVNLLKDDLHPSFPEVSFFHSEENKAPESEELEMEFEDMENVPLDRSSHCQREEAGGRDGGSSLGFKRSYEEHIPYTHMNGGKKNGRILTLPRSNPS
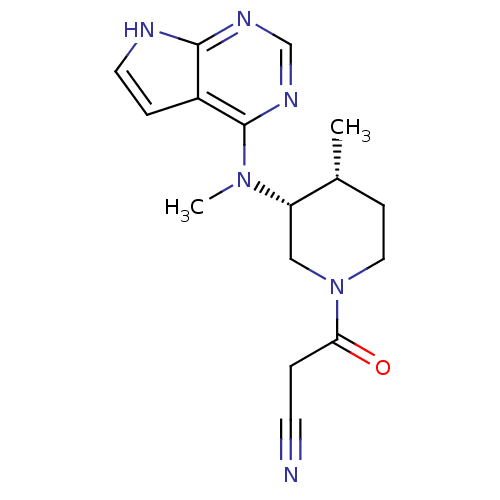
Inhibitor
Name:
BDBM50193995
Synonyms:
3-((3R,4R)-4-methyl-3-(methyl(7H-pyrrolo[2,3-d]pyrimidin-4-yl)amino)piperidin-1-yl)-3-oxopropanenitrile | 3-(4-methyl-3-(methyl(7H-pyrrolo[2,3-d]pyrimidin-4-yl)amino)piperidin-1-yl)-3-oxopropanenitrile | 3-{(3R,4R)-4-methyl-3-[methyl(7H-pyrrolo[2,3-d]pyrimidin-4-yl)amino]piperidin-1-yl}-3-oxopropanenitrile | CHEMBL221959 | CP-690550 | TOFACITINIB CITRATE | Tofacitinib | Tofacitinib citrate (1) | US10112907, Example 00035 | US10399979, Compound Tofacitinib | US10766894, Compound TABLE 1.20 | US10875847, Compound Tofacitinib | US11078206, Example Tofacitinib | US11203595, TABLE 1.20 | US11339167, Example Tofacitinib
Type:
Small organic molecule
Emp. Form.:
C16H20N6O
Mol. Mass.:
312.3696
SMILES:
C[C@@H]1CCN(C[C@@H]1N(C)c1ncnc2[nH]ccc12)C(=O)CC#N |r|