Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
5-hydroxytryptamine receptor 2C
Ligand
BDBM50100425
Substrate
n/a
Meas. Tech.
ChEMBL_1621579 (CHEMBL3863862)
Ki
6400±n/a nM
Citation
 Tosh, DK; Ciancetta, A; Warnick, E; Crane, S; Gao, ZG; Jacobson, KA Structure-Based Scaffold Repurposing for G Protein-Coupled Receptors: Transformation of Adenosine Derivatives into 5HT J Med Chem 59:11006-11026 (2016) [PubMed] Article
Tosh, DK; Ciancetta, A; Warnick, E; Crane, S; Gao, ZG; Jacobson, KA Structure-Based Scaffold Repurposing for G Protein-Coupled Receptors: Transformation of Adenosine Derivatives into 5HT J Med Chem 59:11006-11026 (2016) [PubMed] Article More Info.:
Target
Name:
5-hydroxytryptamine receptor 2C
Synonyms:
5-HT-2C | 5-HT2C | 5-HT2C-INI | 5-HT2c VGI | 5-HTR2C | 5-hydroxytryptamine receptor 1C | 5-hydroxytryptamine receptor 2C (5-HT-2C) | 5-hydroxytryptamine receptor 2C (5HT-2C) | 5HT-1C | 5HT2C_HUMAN | HTR1C | HTR2C | Serotonin (5-HT3) receptor | Serotonin 2c (5-HT2c) receptor | Serotonin Receptor 2C
Type:
G Protein-Coupled Receptor (GPCR)
Mol. Mass.:
51836.79
Organism:
Homo sapiens (Human)
Description:
P28335
Residue:
458
Sequence:
MVNLRNAVHSFLVHLIGLLVWQCDISVSPVAAIVTDIFNTSDGGRFKFPDGVQNWPALSIVIIIIMTIGGNILVIMAVSMEKKLHNATNYFLMSLAIADMLVGLLVMPLSLLAILYDYVWPLPRYLCPVWISLDVLFSTASIMHLCAISLDRYVAIRNPIEHSRFNSRTKAIMKIAIVWAISIGVSVPIPVIGLRDEEKVFVNNTTCVLNDPNFVLIGSFVAFFIPLTIMVITYCLTIYVLRRQALMLLHGHTEEPPGLSLDFLKCCKRNTAEEENSANPNQDQNARRRKKKERRPRGTMQAINNERKASKVLGIVFFVFLIMWCPFFITNILSVLCEKSCNQKLMEKLLNVFVWIGYVCSGINPLVYTLFNKIYRRAFSNYLRCNYKVEKKPPVRQIPRVAATALSGRELNVNIYRHTNEPVIEKASDNEPGIEMQVENLELPVNPSSVVSERISSV
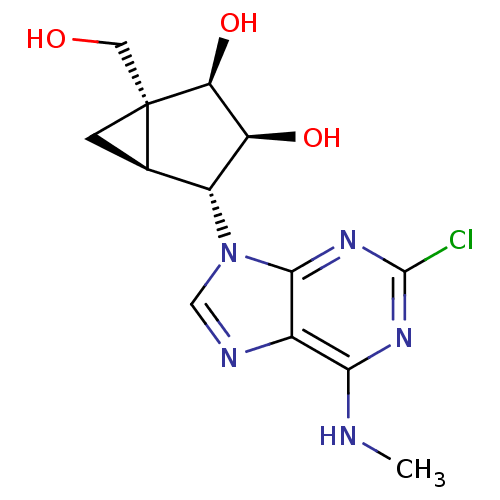
Inhibitor
Name:
BDBM50100425
Synonyms:
(1R,2R,3S,4R,5S)-4-(2-Chloro-6-methylamino-purin-9-yl)-1-hydroxymethyl-bicyclo[3.1.0]hexane-2,3-diol | CHEMBL286739
Type:
Small organic molecule
Emp. Form.:
C13H16ClN5O3
Mol. Mass.:
325.751
SMILES:
CNc1nc(Cl)nc2n(cnc12)[C@@H]1[C@H]2C[C@@]2(CO)[C@@H](O)[C@H]1O