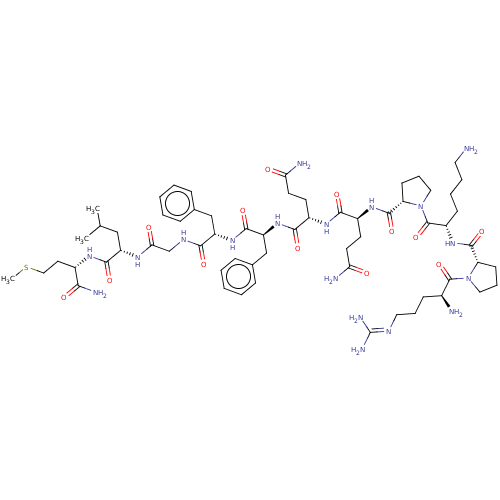
Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Substance-P receptor
Ligand
BDBM50001450
Substrate
n/a
Meas. Tech.
ChEMBL_1632341 (CHEMBL3875047)
IC50
1.1±n/a nM
Citation
 Dyniewicz, J; Lipinski, PF; Kosson, P; Lesniak, A; Bochynska-Czyz, M; Muchowska, A; Tourw�, D; Ballet, S; Misicka, A; Lipkowski, AW Hydrazone Linker as a Useful Tool for Preparing Chimeric Peptide/Nonpeptide Bifunctional Compounds. ACS Med Chem Lett 8:73-77 (2017) [PubMed] Article
Dyniewicz, J; Lipinski, PF; Kosson, P; Lesniak, A; Bochynska-Czyz, M; Muchowska, A; Tourw�, D; Ballet, S; Misicka, A; Lipkowski, AW Hydrazone Linker as a Useful Tool for Preparing Chimeric Peptide/Nonpeptide Bifunctional Compounds. ACS Med Chem Lett 8:73-77 (2017) [PubMed] Article More Info.:
Target
Name:
Substance-P receptor
Synonyms:
NK-1 receptor | NK-1R | NK1 Receptor | NK1R_RAT | Neurokinin 1 receptor | Neurokinin NK1 | SPR | Substance-P receptor | Tac1r | Tachykinin receptor 1 | Tacr1
Type:
G Protein-Coupled Receptor (GPCR)
Mol. Mass.:
46371.54
Organism:
Rattus norvegicus (rat)
Description:
Competition binding assays were carried out using membrane preparations from transfected CHO cells that constitutively expressed the rat NK1 receptor.
Residue:
407
Sequence:
MDNVLPMDSDLFPNISTNTSESNQFVQPTWQIVLWAAAYTVIVVTSVVGNVVVIWIILAHKRMRTVTNYFLVNLAFAEACMAAFNTVVNFTYAVHNVWYYGLFYCKFHNFFPIAALFASIYSMTAVAFDRYMAIIHPLQPRLSATATKVVIFVIWVLALLLAFPQGYYSTTETMPSRVVCMIEWPEHPNRTYEKAYHICVTVLIYFLPLLVIGYAYTVVGITLWASEIPGDSSDRYHEQVSAKRKVVKMMIVVVCTFAICWLPFHVFFLLPYINPDLYLKKFIQQVYLASMWLAMSSTMYNPIIYCCLNDRFRLGFKHAFRCCPFISAGDYEGLEMKSTRYLQTQSSVYKVSRLETTISTVVGAHEEEPEEGPKATPSSLDLTSNGSSRSNSKTMTESSSFYSNMLA
Inhibitor
Name:
BDBM50001450
Synonyms:
(SP)Arg-Pro-Lys-Pro-Gln-Gln-Phe-Phe-Gly-Leu-Met-NH2 | Arg-Pro-Lys-Pro-Gln-Gln-Phe-Phe-Gly-Leu-Met NH2 | Arg-Pro-Lys-Pro-Gln-Gln-Phe-Phe-Gly-Leu-Met-NH2 | Arg-Pro-Lys-Pro-Gln-Gln-Phe-Phe-Gly-Leu-Met-NH2(Substance P) | Arg-Pro-Lys-Pro-Gln-Gln-Phe-Phe-Gly-Leu-Met-NH2.(Substance P) | Arg-Pro-Lys-Pro-Gln-Gln-Phe-Phe-Gly-Leu-Met-amine | ArgProLysProGlnGlnPhePheGlyLeuMet | CHEMBL235363 | H-Arg-Pro-Lys-Pro-Gln-Gln-Phe-D-Phe-Gly-Leu-Met-NH2 | H-Arg-Pro-Lys-Pro-Gln-Gln-Phe-Phe-Gly-Leu-Met-NH2(Substance P) | Substance P | Substance P (Arg-Pro-Lys-Pro-Gln-Gln-Phe-Phe-Gly-Leu-MetNH2) | Substance P analogue | tachykinin substance P (SP)
Type:
Small organic molecule
Emp. Form.:
C63H98N18O13S
Mol. Mass.:
1347.63
SMILES:
[#6]-[#16]-[#6]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#6])-[#6])-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccccc1)-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccccc1)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@@H](-[#7])-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](-[#7])=O |r|