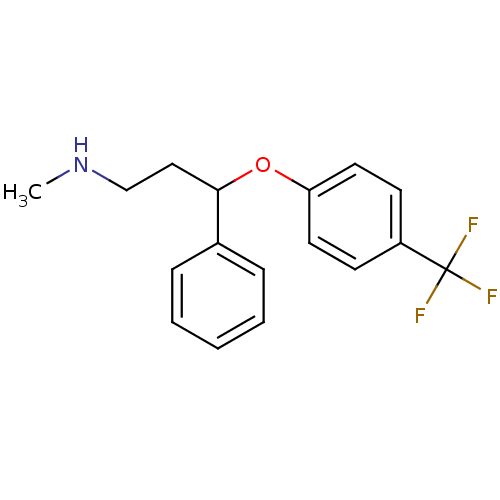
BDBM30130 CHEMBL1201082::CHEMBL41::Fluoxetin::Fluoxetine::Prozac::US9120771, Fluoxetine::cid_62857
SMILES CNCCC(Oc1ccc(cc1)C(F)(F)F)c1ccccc1
InChI Key InChIKey=RTHCYVBBDHJXIQ-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 10 hits for monomerid = 30130
Found 10 hits for monomerid = 30130
Target5-hydroxytryptamine receptor 2A(Rattus norvegicus (rat))
Emory University
Curated by PDSP Ki Database
Emory University
Curated by PDSP Ki Database
Target5-hydroxytryptamine receptor 2A(Rattus norvegicus (rat))
Emory University
Curated by PDSP Ki Database
Emory University
Curated by PDSP Ki Database
Target5-hydroxytryptamine receptor 2A(Homo sapiens (Human))
Roche Bioscience
Curated by PDSP Ki Database
Roche Bioscience
Curated by PDSP Ki Database
Target5-hydroxytryptamine receptor 2A(Homo sapiens (Human))
Roche Bioscience
Curated by PDSP Ki Database
Roche Bioscience
Curated by PDSP Ki Database
Target5-hydroxytryptamine receptor 2A(Rattus norvegicus (rat))
Emory University
Curated by PDSP Ki Database
Emory University
Curated by PDSP Ki Database
Target5-hydroxytryptamine receptor 2A(Rattus norvegicus (rat))
Emory University
Curated by PDSP Ki Database
Emory University
Curated by PDSP Ki Database
Target5-hydroxytryptamine receptor 2A(Rattus norvegicus (rat))
Emory University
Curated by PDSP Ki Database
Emory University
Curated by PDSP Ki Database
Target5-hydroxytryptamine receptor 2A(Rattus norvegicus (rat))
Emory University
Curated by PDSP Ki Database
Emory University
Curated by PDSP Ki Database
Target5-hydroxytryptamine receptor 2A(Rattus norvegicus (rat))
Emory University
Curated by PDSP Ki Database
Emory University
Curated by PDSP Ki Database
Target5-hydroxytryptamine receptor 2A(Homo sapiens (Human))
Roche Bioscience
Curated by PDSP Ki Database
Roche Bioscience
Curated by PDSP Ki Database
Affinity DataIC50: 710nMAssay Description:Displacement of [3H]ketanserin from 5HT2A receptor (unknown origin) by cell based radioligand competitive binding analysisMore data for this Ligand-Target Pair