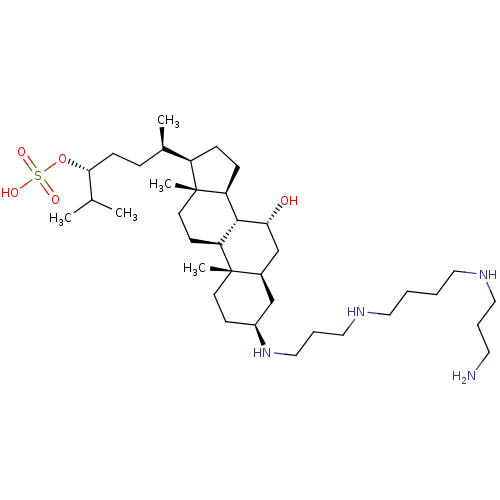
BDBM50333649 (3R,6R)-6-((3S,5R,7R,8R,9S,10S,13R,14S,17R)-3-(3-(4-(3-aminopropylamino)butylamino)propylamino)-7-hydroxy-10,13-dimethyl-hexadecahydro-1H-cyclopenta[a]phenanthren-17-yl)-2-methylheptan-3-yl hydrogen sulfate::CHEMBL508583::trodusquemine
SMILES CC(C)[C@@H](CC[C@@H](C)[C@H]1CC[C@H]2[C@@H]3[C@H](O)C[C@H]4C[C@H](CC[C@]4(C)[C@H]3CC[C@]12C)NCCCNCCCCNCCCN)OS(O)(=O)=O
InChI Key InChIKey=WUJVPODXELZABP-FWJXURDUSA-N
Data 5 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50333649
Found 5 hits for monomerid = 50333649
TargetTyrosine-protein phosphatase non-receptor type 1(Homo sapiens (Human))
Sanford-Burnham Medical Research Institute
Curated by ChEMBL
Sanford-Burnham Medical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibition of full length PTP1B (unknown origin) after 30 mins by ELISAMore data for this Ligand-Target Pair
TargetTyrosine-protein phosphatase non-receptor type 1(Homo sapiens (Human))
Sanford-Burnham Medical Research Institute
Curated by ChEMBL
Sanford-Burnham Medical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibition of recombinant PTP1BMore data for this Ligand-Target Pair
TargetTyrosine-protein phosphatase non-receptor type 1(Homo sapiens (Human))
Sanford-Burnham Medical Research Institute
Curated by ChEMBL
Sanford-Burnham Medical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibition of human recombinant PTP1B (1 to 322 residues) expressed in Escherichia coli using pNPP as substrate preincubated with substrate for 10 mi...More data for this Ligand-Target Pair
TargetTyrosine-protein phosphatase non-receptor type 1(Homo sapiens (Human))
Sanford-Burnham Medical Research Institute
Curated by ChEMBL
Sanford-Burnham Medical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibition of full length PTP1B (unknown origin) by ELISAMore data for this Ligand-Target Pair
TargetTyrosine-protein phosphatase non-receptor type 1(Homo sapiens (Human))
Sanford-Burnham Medical Research Institute
Curated by ChEMBL
Sanford-Burnham Medical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.30E+3nMAssay Description:Inhibition of PTP1BMore data for this Ligand-Target Pair