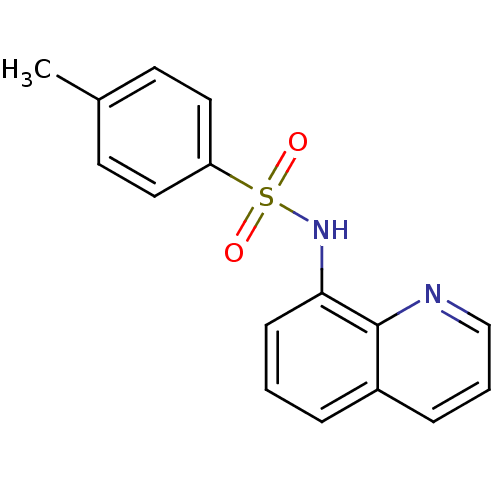
BDBM60223 4-methyl-N-(8-quinolinyl)benzenesulfonamide::4-methyl-N-(8-quinolyl)benzenesulfonamide::4-methyl-N-8-quinolinylbenzenesulfonamide::4-methyl-N-quinolin-8-yl-benzenesulfonamide::4-methyl-N-quinolin-8-ylbenzenesulfonamide::MLS000530172::SMR000135149::cid_82533
SMILES Cc1ccc(cc1)S(=O)(=O)Nc1cccc2cccnc12
InChI Key InChIKey=ZSMKPYXVUIWTCT-UHFFFAOYSA-N
Data 4 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 60223
Found 4 hits for monomerid = 60223
TargetEndothelial PAS domain-containing protein 1(Human)
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
TargetKappa-type opioid receptor(Human)
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 2.57E+3nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor(Human)
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 3.67E+3nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute(SBMRI, San Diego, C...More data for this Ligand-Target Pair
Affinity DataIC50: 2.66E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford- Sanford-Burnham Medical Research Institute(SBMRI, San...More data for this Ligand-Target Pair