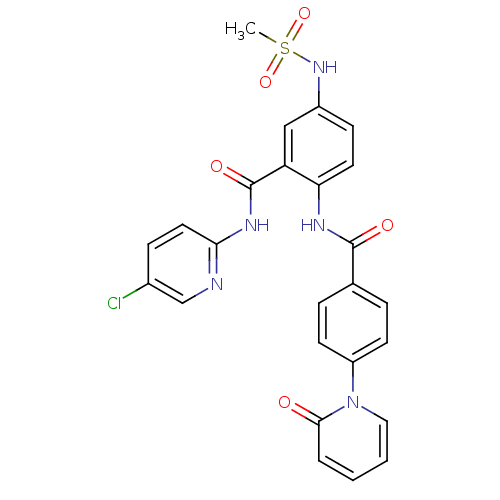
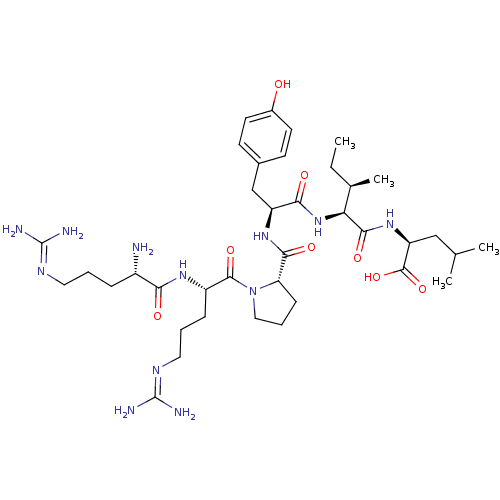
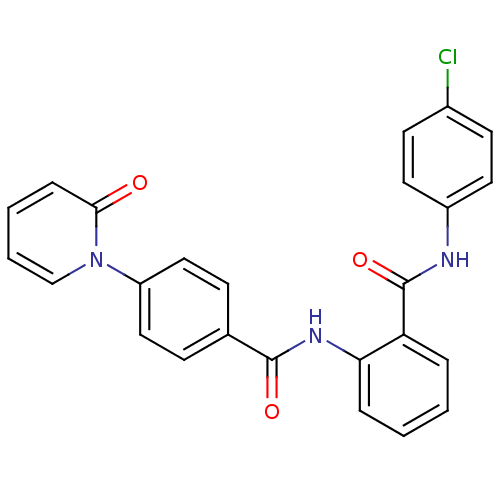
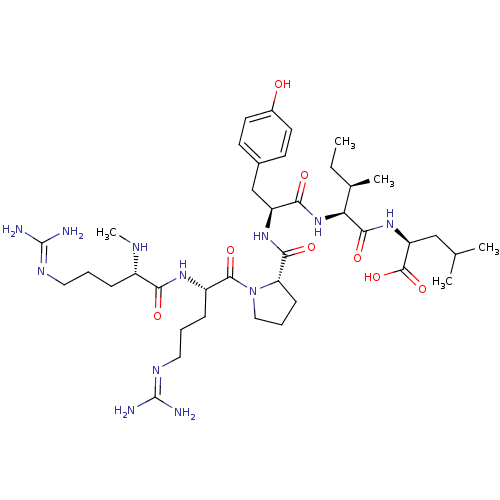
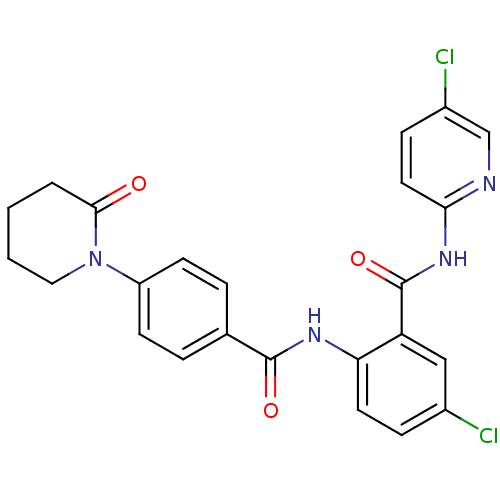
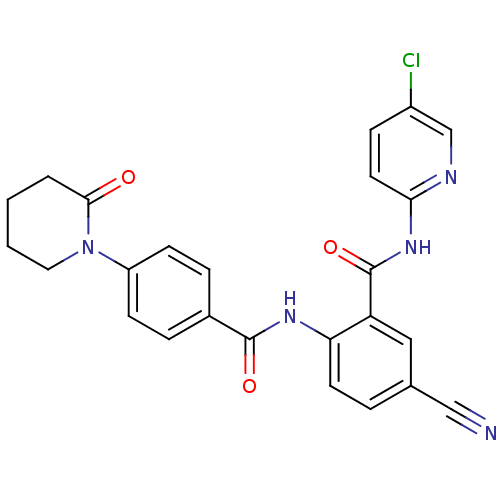
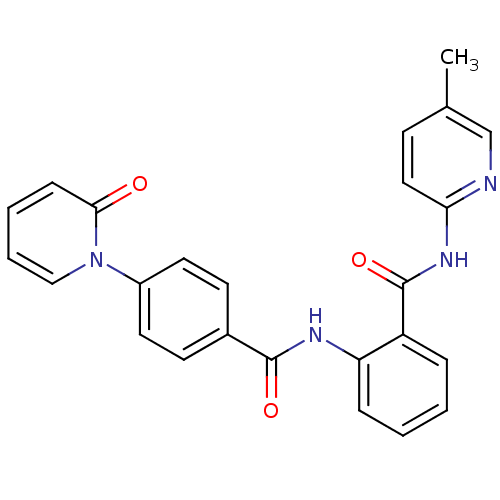
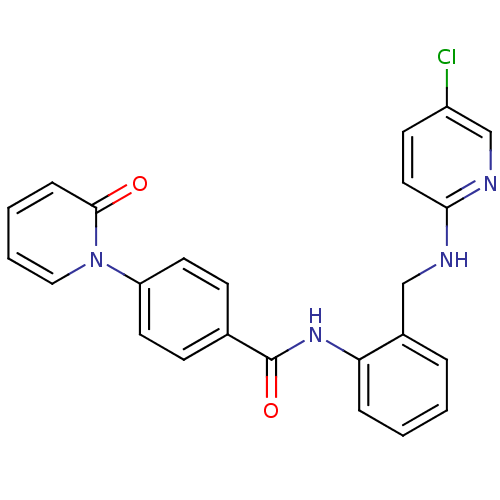
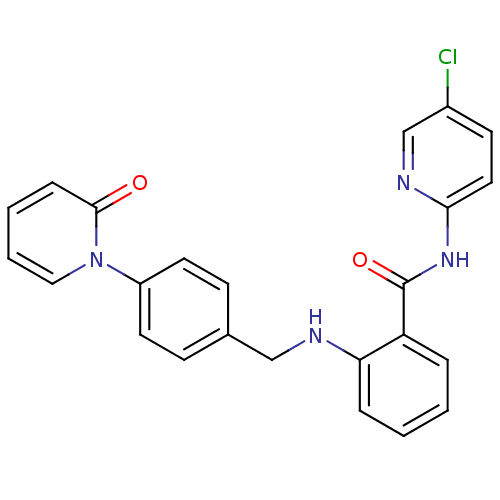
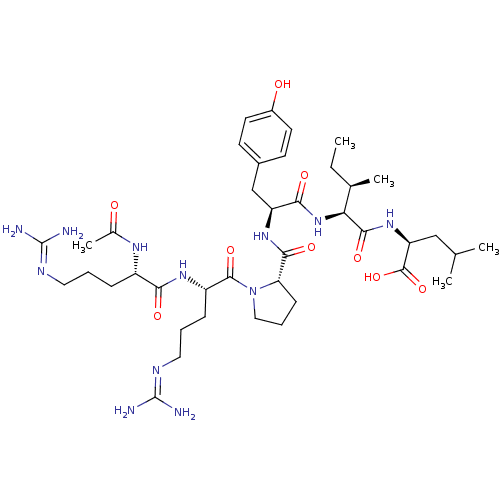
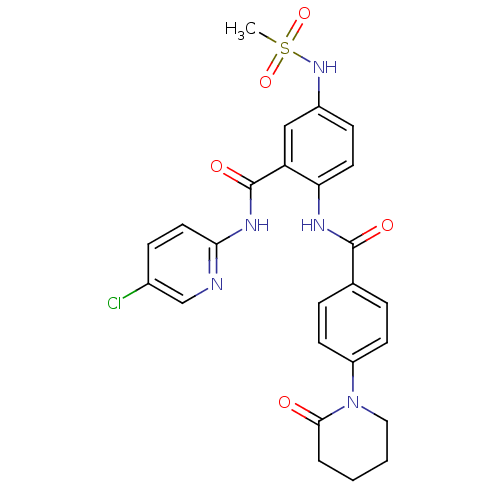
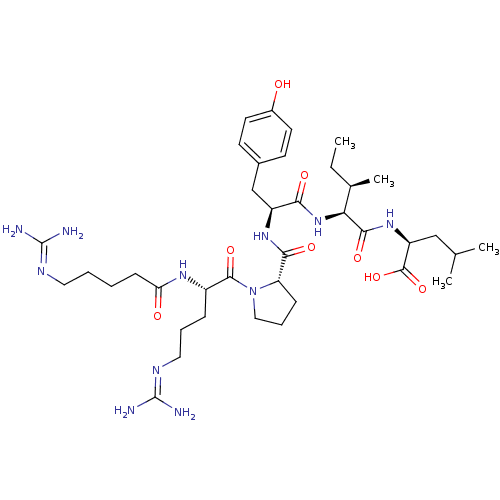
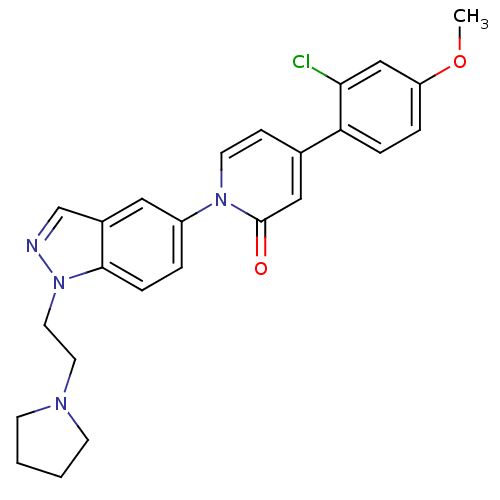
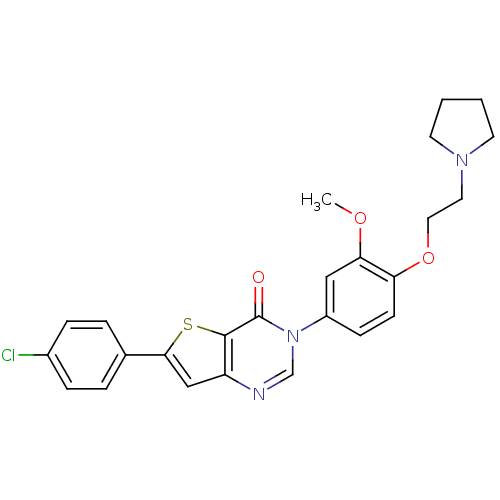
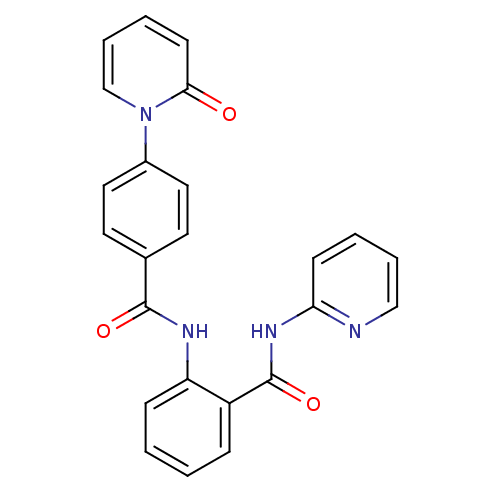
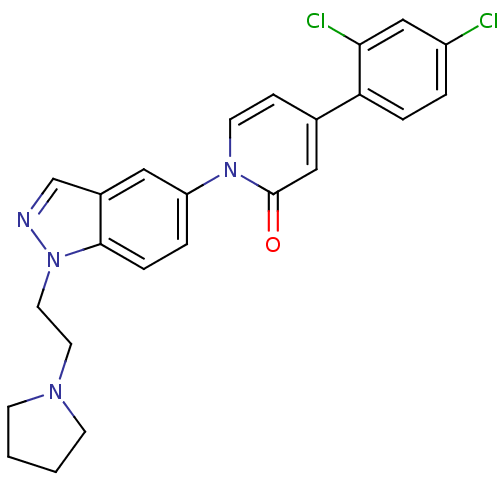
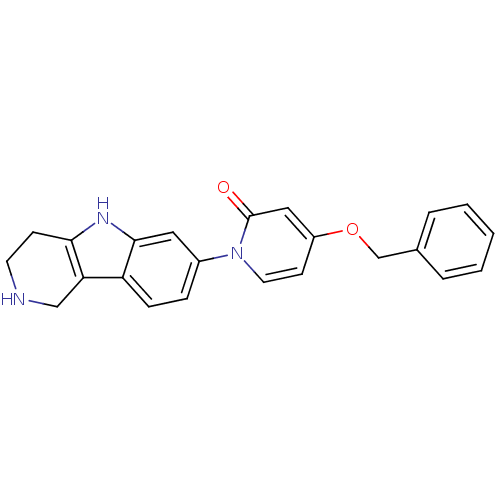
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.00500nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.0130nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.0280nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.0470nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.0570nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.0600nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.0650nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.0800nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.120nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.140nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.140nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.140nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetNeurotensin receptor type 1(Homo sapiens (Human))
Medical University Of South Carolina
Curated by ChEMBL
Medical University Of South Carolina
Curated by ChEMBL
Affinity DataKi: 0.140nMAssay Description:Binding affinity to human NTR1More data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.190nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.210nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.240nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetNeurotensin receptor type 1(Homo sapiens (Human))
Medical University Of South Carolina
Curated by ChEMBL
Medical University Of South Carolina
Curated by ChEMBL
Affinity DataKi: 0.290nMAssay Description:Displacement of [125I]I-Tyr(3)NT from human NTR1More data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.330nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.450nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.580nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.620nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetNeurotensin receptor type 1(Homo sapiens (Human))
Medical University Of South Carolina
Curated by ChEMBL
Medical University Of South Carolina
Curated by ChEMBL
Affinity DataKi: 0.730nMAssay Description:Displacement of [125I]I-Tyr(3)NT from human NTR1More data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 1nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetNeurotensin receptor type 1(Homo sapiens (Human))
Medical University Of South Carolina
Curated by ChEMBL
Medical University Of South Carolina
Curated by ChEMBL
Affinity DataKi: 1.05nMAssay Description:Displacement of [125I]I-Tyr(3)NT from human NTR1More data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 1.10nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 1.20nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
Affinity DataKi: 1.20nMAssay Description:Displacement of [3H]GW803430 from MCH-1 receptor expressed in CHO-K1 cellsMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 1.30nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetNeurotensin receptor type 1(Homo sapiens (Human))
Medical University Of South Carolina
Curated by ChEMBL
Medical University Of South Carolina
Curated by ChEMBL
Affinity DataKi: 1.64nMAssay Description:Displacement of [125I]I-Tyr(3)NT from human NTR1More data for this Ligand-Target Pair
TargetNeurotensin receptor type 1(Homo sapiens (Human))
Medical University Of South Carolina
Curated by ChEMBL
Medical University Of South Carolina
Curated by ChEMBL
Affinity DataKi: 1.81nMAssay Description:Displacement of [125I]I-Tyr(3)NT from human NTR1More data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 2nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
TargetNeurotensin receptor type 1(Homo sapiens (Human))
Medical University Of South Carolina
Curated by ChEMBL
Medical University Of South Carolina
Curated by ChEMBL
Affinity DataKi: 2.01nMAssay Description:Displacement of [125I]I-Tyr(3)NT from human NTR1More data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 2.20nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
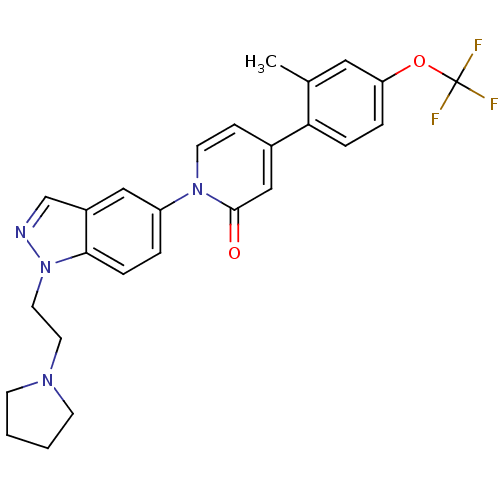
Affinity DataKi: 2.40nMAssay Description:Displacement of [3H]4-(benzyloxy)-1-(1-(2-(pyrrolidin-1-yl)ethyl)-1H-indazol-5-yl)pyridin-2(1H)-one from MCH-1 receptor expressed in CHO-K1 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 2.60nMAssay Description:Displacement of [3H]4-(benzyloxy)-1-(1-(2-(pyrrolidin-1-yl)ethyl)-1H-indazol-5-yl)pyridin-2(1H)-one from MCH-1 receptor expressed in CHO-K1 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 2.60nMAssay Description:Displacement of [3H]4-(benzyloxy)-1-(1-(2-(pyrrolidin-1-yl)ethyl)-1H-indazol-5-yl)pyridin-2(1H)-one from MCH-1 receptor expressed in CHO-K1 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 2.60nMAssay Description:Binding affinity to human recombinant G9a catalytic domain amino acid 913 to 1193 expressed in Escherichia coli BL21 (DE3) by isothermal titration ca...More data for this Ligand-Target Pair
Affinity DataKi: 2.60nMAssay Description:Displacement of [3H]4-(benzyloxy)-1-(1-(2-(pyrrolidin-1-yl)ethyl)-1H-indazol-5-yl)pyridin-2(1H)-one from MCH-1 receptor expressed in CHO-K1 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 2.60nMAssay Description:Displacement of [3H]GW803430 from MCH-1 receptor expressed in CHO-K1 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 2.70nMAssay Description:Displacement of [3H]4-(benzyloxy)-1-(1-(2-(pyrrolidin-1-yl)ethyl)-1H-indazol-5-yl)pyridin-2(1H)-one from MCH-1 receptor expressed in CHO-K1 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 2.80nMAssay Description:Displacement of [3H]GW803430 from MCH-1 receptor expressed in CHO-K1 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 2.80nMAssay Description:Displacement of [3H]GW803430 from MCH-1 receptor expressed in CHO-K1 cellsMore data for this Ligand-Target Pair
TargetNeurotensin receptor type 1(Homo sapiens (Human))
Medical University Of South Carolina
Curated by ChEMBL
Medical University Of South Carolina
Curated by ChEMBL
Affinity DataKi: 2.99nMAssay Description:Displacement of [125I]I-Tyr(3)NT from human NTR1More data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 3nMAssay Description:Inhibition of human factor 10aMore data for this Ligand-Target Pair
Affinity DataKi: 3.10nMAssay Description:Displacement of [3H]4-(benzyloxy)-1-(1-(2-(pyrrolidin-1-yl)ethyl)-1H-indazol-5-yl)pyridin-2(1H)-one from MCH-1 receptor expressed in CHO-K1 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 3.40nMAssay Description:Displacement of [3H]GW803430 from MCH-1 receptor expressed in CHO-K1 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 3.60nMAssay Description:Displacement of [3H]4-(benzyloxy)-1-(1-(2-(pyrrolidin-1-yl)ethyl)-1H-indazol-5-yl)pyridin-2(1H)-one from MCH-1 receptor expressed in CHO-K1 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 3.70nMAssay Description:Competitive inhibition of G9a by fluorescence polarization assay in presence of fluorescein-labeled H3 peptideMore data for this Ligand-Target Pair
Affinity DataKi: 3.80nMAssay Description:Displacement of [3H]4-(benzyloxy)-1-(1-(2-(pyrrolidin-1-yl)ethyl)-1H-indazol-5-yl)pyridin-2(1H)-one from MCH-1 receptor expressed in CHO-K1 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 3.90nMAssay Description:Displacement of [3H]GW803430 from MCH-1 receptor expressed in CHO-K1 cellsMore data for this Ligand-Target Pair


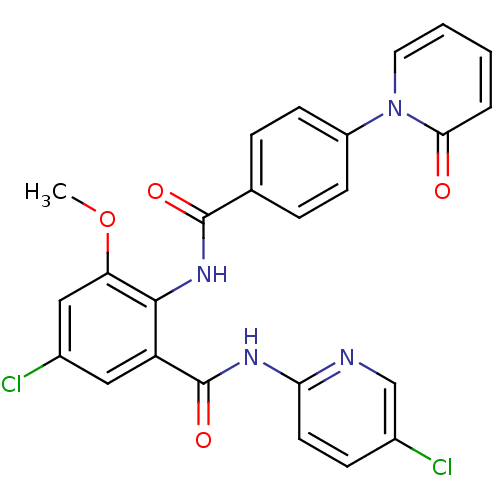
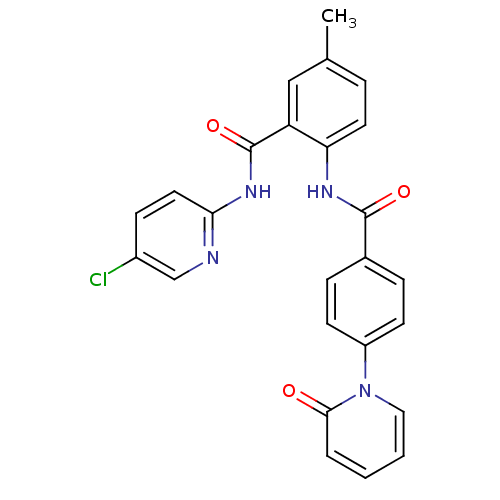

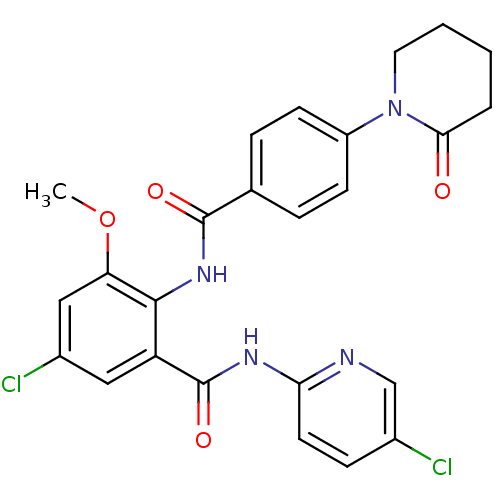
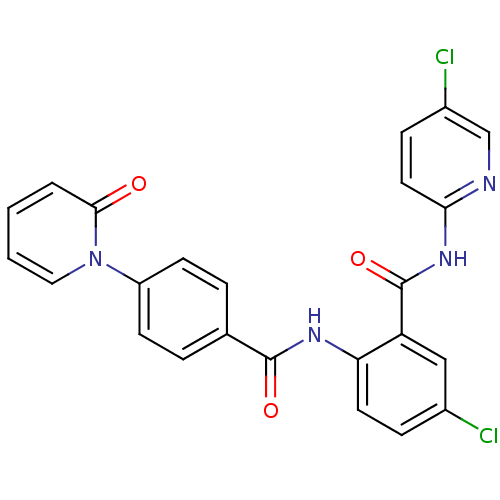
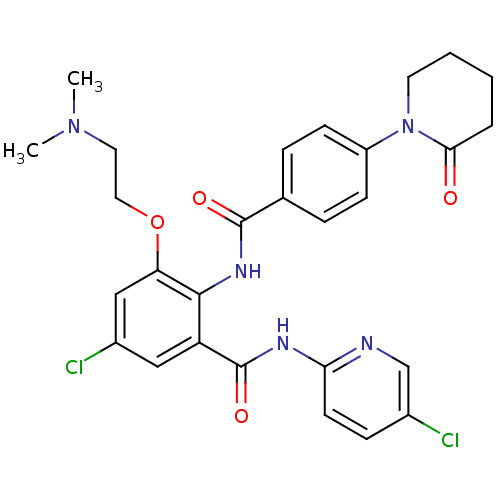

 3D Structure (crystal)
3D Structure (crystal)