TargetAngiotensin-converting enzyme(Homo sapiens (Human))
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Affinity DataIC50: 0.100nMAssay Description:Inhibitory activity against Angiotensin I converting enzyme (ACE) from human blood serumMore data for this Ligand-Target Pair
TargetAngiotensin-converting enzyme(Homo sapiens (Human))
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Affinity DataIC50: 0.100nMAssay Description:Inhibition of human serum ACEMore data for this Ligand-Target Pair
TargetAngiotensin-converting enzyme(Homo sapiens (Human))
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Affinity DataIC50: 0.100nMAssay Description:Inhibition of angiotensin converting enzyme (unknown origin)More data for this Ligand-Target Pair
TargetAngiotensin-converting enzyme(Homo sapiens (Human))
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Affinity DataIC50: 1.20nMAssay Description:Inhibition of angiotensin-converting enzyme (unknown origin)More data for this Ligand-Target Pair
TargetAngiotensin-converting enzyme(Homo sapiens (Human))
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Affinity DataIC50: 1.20nMAssay Description:Inhibition of Angiotensin I converting enzyme (ACE) in Bothrops jararaca venomMore data for this Ligand-Target Pair
TargetAngiotensin-converting enzyme(Homo sapiens (Human))
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Affinity DataIC50: 1.20nMAssay Description:Inhibition of Angiotensin I converting enzymeMore data for this Ligand-Target Pair
TargetAngiotensin-converting enzyme(Homo sapiens (Human))
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Affinity DataIC50: 1.20nMAssay Description:Inhibitory activity against angiotensin converting enzyme (ACE)More data for this Ligand-Target Pair
TargetAngiotensin-converting enzyme(Homo sapiens (Human))
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Affinity DataIC50: 3.98nMAssay Description:Inhibition of angiotensin I converting enzyme in silicoMore data for this Ligand-Target Pair
TargetAngiotensin-converting enzyme(Homo sapiens (Human))
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Affinity DataIC50: 4.70nMAssay Description:Inhibitory activity against angiotensin I converting enzyme (ACE)Checked by AuthorMore data for this Ligand-Target Pair
TargetAngiotensin-converting enzyme(Homo sapiens (Human))
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Institute Of Biomedical Chemistry Of Russian Academy Of Medical Sciences
Curated by ChEMBL
Affinity DataIC50: 7.90nMAssay Description:Inhibition of ACE (unknown origin) using Hippuryl-L-Histidyl-L-Leucine as substrate after 60 mins by colorimetric methodMore data for this Ligand-Target Pair
TargetAngiotensin-converting enzyme(Oryctolagus cuniculus)
Csir-Indian Institute Of Chemical Technology
Curated by ChEMBL
Csir-Indian Institute Of Chemical Technology
Curated by ChEMBL
Affinity DataIC50: 262nMAssay Description:Inhibition of rabbit lung ACE assessed as hydrolysis of hippuryl-histidyl-leucine to hippuric acid and histidyl-leucine after 30 minsMore data for this Ligand-Target Pair
TargetAngiotensin-converting enzyme(Oryctolagus cuniculus)
Csir-Indian Institute Of Chemical Technology
Curated by ChEMBL
Csir-Indian Institute Of Chemical Technology
Curated by ChEMBL
Affinity DataIC50: 281nMAssay Description:Inhibition of rabbit lung ACE using hippuryl-histidyl-leucine as substrate assessed as release of hippuric acid incubated for 10 mins prior to substr...More data for this Ligand-Target Pair
TargetAngiotensin-converting enzyme(Oryctolagus cuniculus)
Csir-Indian Institute Of Chemical Technology
Curated by ChEMBL
Csir-Indian Institute Of Chemical Technology
Curated by ChEMBL
Affinity DataIC50: 318nMAssay Description:Inhibition of rabbit lung ACE assessed as hippuryl-histidyl-leucine hydrolysis after 30 mins by colorimetric methodMore data for this Ligand-Target Pair
Affinity DataIC50: >1.33E+5nMAssay Description:Inhibition of human MRP2 overexpressed in Sf9 cell membrane vesicles assessed as uptake of [3H]-estradiol-17beta-D-glucuronide in presence of ATP and...More data for this Ligand-Target Pair
Affinity DataIC50: >1.33E+5nMAssay Description:Inhibition of human MRP3 overexpressed in Sf9 insect cell membrane vesicles assessed as uptake of [3H]-estradiol-17beta-D-glucuronide in presence of ...More data for this Ligand-Target Pair
Affinity DataIC50: >1.33E+5nMAssay Description:Inhibition of human MRP4 overexpressed in Sf9 cell membrane vesicles assessed as uptake of [3H]-estradiol-17beta-D-glucuronide in presence of ATP and...More data for this Ligand-Target Pair
Affinity DataIC50: >1.33E+5nMAssay Description:Inhibition of human BSEP overexpressed in Sf9 cell membrane vesicles assessed as uptake of [3H]-taurocholate in presence of ATP measured after 15 to ...More data for this Ligand-Target Pair

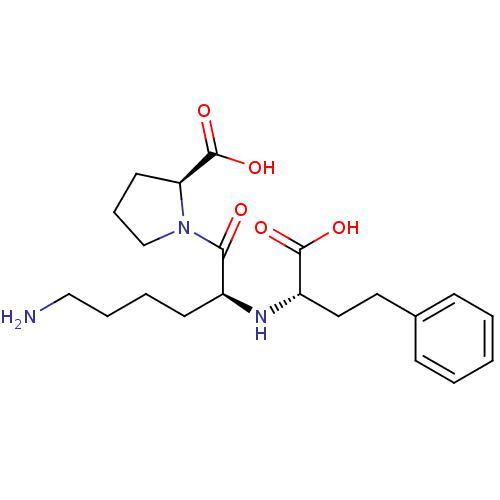
 3D Structure (crystal)
3D Structure (crystal)