Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Non-receptor tyrosine-protein kinase TYK2
Ligand
BDBM50193995
Substrate
n/a
Meas. Tech.
ChEBML_1682728
IC50
12±n/a nM
Citation
 Siu, T; Brubaker, J; Fuller, P; Torres, L; Zeng, H; Close, J; Mampreian, DM; Shi, F; Liu, D; Fradera, X; Johnson, K; Bays, N; Kadic, E; He, F; Goldenblatt, P; Shaffer, L; Patel, SB; Lesburg, CA; Alpert, C; Dorosh, L; Deshmukh, SV; Yu, H; Klappenbach, J; Elwood, F; Dinsmore, CJ; Fernandez, R; Moy, L; Young, JR The Discovery of 3-((4-Chloro-3-methoxyphenyl)amino)-1-((3R,4S)-4-cyanotetrahydro-2H-pyran-3-yl)-1H-pyrazole-4-carboxamide, a Highly Ligand Efficient and Efficacious Janus Kinase 1 Selective Inhibitor with Favorable Pharmacokinetic Properties. J Med Chem 60:9676-9690 (2017) [PubMed] Article
Siu, T; Brubaker, J; Fuller, P; Torres, L; Zeng, H; Close, J; Mampreian, DM; Shi, F; Liu, D; Fradera, X; Johnson, K; Bays, N; Kadic, E; He, F; Goldenblatt, P; Shaffer, L; Patel, SB; Lesburg, CA; Alpert, C; Dorosh, L; Deshmukh, SV; Yu, H; Klappenbach, J; Elwood, F; Dinsmore, CJ; Fernandez, R; Moy, L; Young, JR The Discovery of 3-((4-Chloro-3-methoxyphenyl)amino)-1-((3R,4S)-4-cyanotetrahydro-2H-pyran-3-yl)-1H-pyrazole-4-carboxamide, a Highly Ligand Efficient and Efficacious Janus Kinase 1 Selective Inhibitor with Favorable Pharmacokinetic Properties. J Med Chem 60:9676-9690 (2017) [PubMed] Article More Info.:
Target
Name:
Non-receptor tyrosine-protein kinase TYK2
Synonyms:
Non-receptor tyrosine-protein kinase TYK2 (TYK2) | TYK2 | TYK2_HUMAN | Tyrosine-protein kinase TYK2
Type:
Protein
Mol. Mass.:
133666.03
Organism:
Homo sapiens (Human)
Description:
P29597
Residue:
1187
Sequence:
MPLRHWGMARGSKPVGDGAQPMAAMGGLKVLLHWAGPGGGEPWVTFSESSLTAEEVCIHIAHKVGITPPCFNLFALFDAQAQVWLPPNHILEIPRDASLMLYFRIRFYFRNWHGMNPREPAVYRCGPPGTEASSDQTAQGMQLLDPASFEYLFEQGKHEFVNDVASLWELSTEEEIHHFKNESLGMAFLHLCHLALRHGIPLEEVAKKTSFKDCIPRSFRRHIRQHSALTRLRLRNVFRRFLRDFQPGRLSQQMVMVKYLATLERLAPRFGTERVPVCHLRLLAQAEGEPCYIRDSGVAPTDPGPESAAGPPTHEVLVTGTGGIQWWPVEEEVNKEEGSSGSSGRNPQASLFGKKAKAHKAVGQPADRPREPLWAYFCDFRDITHVVLKEHCVSIHRQDNKCLELSLPSRAAALSFVSLVDGYFRLTADSSHYLCHEVAPPRLVMSIRDGIHGPLLEPFVQAKLRPEDGLYLIHWSTSHPYRLILTVAQRSQAPDGMQSLRLRKFPIEQQDGAFVLEGWGRSFPSVRELGAALQGCLLRAGDDCFSLRRCCLPQPGETSNLIIMRGARASPRTLNLSQLSFHRVDQKEITQLSHLGQGTRTNVYEGRLRVEGSGDPEEGKMDDEDPLVPGRDRGQELRVVLKVLDPSHHDIALAFYETASLMSQVSHTHLAFVHGVCVRGPENIMVTEYVEHGPLDVWLRRERGHVPMAWKMVVAQQLASALSYLENKNLVHGNVCGRNILLARLGLAEGTSPFIKLSDPGVGLGALSREERVERIPWLAPECLPGGANSLSTAMDKWGFGATLLEICFDGEAPLQSRSPSEKEHFYQRQHRLPEPSCPQLATLTSQCLTYEPTQRPSFRTILRDLTRLQPHNLADVLTVNPDSPASDPTVFHKRYLKKIRDLGEGHFGKVSLYCYDPTNDGTGEMVAVKALKADCGPQHRSGWKQEIDILRTLYHEHIIKYKGCCEDQGEKSLQLVMEYVPLGSLRDYLPRHSIGLAQLLLFAQQICEGMAYLHAQHYIHRDLAARNVLLDNDRLVKIGDFGLAKAVPEGHEYYRVREDGDSPVFWYAPECLKEYKFYYASDVWSFGVTLYELLTHCDSSQSPPTKFLELIGIAQGQMTVLRLTELLERGERLPRPDKCPCEVYHLMKNCWETEASFRPTFENLIPILKTVHEKYQGQAPSVFSVC
Inhibitor
Name:
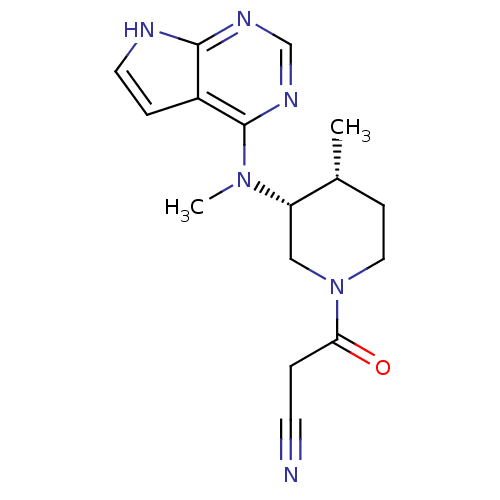
BDBM50193995
Synonyms:
3-((3R,4R)-4-methyl-3-(methyl(7H-pyrrolo[2,3-d]pyrimidin-4-yl)amino)piperidin-1-yl)-3-oxopropanenitrile | 3-(4-methyl-3-(methyl(7H-pyrrolo[2,3-d]pyrimidin-4-yl)amino)piperidin-1-yl)-3-oxopropanenitrile | 3-{(3R,4R)-4-methyl-3-[methyl(7H-pyrrolo[2,3-d]pyrimidin-4-yl)amino]piperidin-1-yl}-3-oxopropanenitrile | CHEMBL221959 | CP-690550 | TOFACITINIB CITRATE | Tofacitinib | Tofacitinib citrate (1) | US10112907, Example 00035 | US10399979, Compound Tofacitinib | US10766894, Compound TABLE 1.20 | US10875847, Compound Tofacitinib | US11078206, Example Tofacitinib | US11203595, TABLE 1.20 | US11339167, Example Tofacitinib
Type:
Small organic molecule
Emp. Form.:
C16H20N6O
Mol. Mass.:
312.3696
SMILES:
C[C@@H]1CCN(C[C@@H]1N(C)c1ncnc2[nH]ccc12)C(=O)CC#N |r|