Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Prothrombin
Ligand
BDBM50072531
Substrate
n/a
Meas. Tech.
ChEMBL_225581 (CHEMBL847937)
Ki
75±n/a nM
Citation
 St-Denis, Y; Augelli-Szafran, CE; Bachand, B; Berryman, KA; DiMaio, J; Doherty, AM; Edmunds, JJ; Leblond, L; Lévesque, S; Narasimhan, LS; Penvose-Yi, JR; Rubin, JR; Tarazi, M; Winocour, PD; Siddiqui, MA Potent bicyclic lactam inhibitors of thrombin: Part I: P3 modifications. Bioorg Med Chem Lett 8:3193-8 (1999) [PubMed] Article
St-Denis, Y; Augelli-Szafran, CE; Bachand, B; Berryman, KA; DiMaio, J; Doherty, AM; Edmunds, JJ; Leblond, L; Lévesque, S; Narasimhan, LS; Penvose-Yi, JR; Rubin, JR; Tarazi, M; Winocour, PD; Siddiqui, MA Potent bicyclic lactam inhibitors of thrombin: Part I: P3 modifications. Bioorg Med Chem Lett 8:3193-8 (1999) [PubMed] Article More Info.:
Target
Name:
Prothrombin
Synonyms:
Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain
Type:
Protein
Mol. Mass.:
70029.57
Organism:
Homo sapiens (Human)
Description:
P00734
Residue:
622
Sequence:
MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLERECVEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHVNITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQECSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASAQAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETGDGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYIDGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTENDLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHPVCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDSTRIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKYGFYTHVFRLKKWIQKVIDQFGE
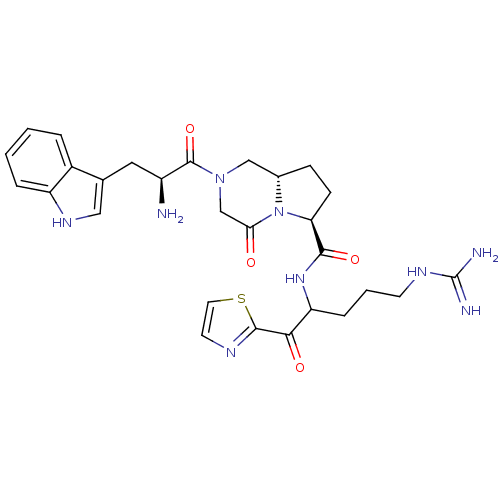
Inhibitor
Name:
BDBM50072531
Synonyms:
(6S,8aS)-2-[(S)-2-Amino-3-(1H-indol-3-yl)-propionyl]-4-oxo-octahydro-pyrrolo[1,2-a]pyrazine-6-carboxylic acid [4-guanidino-1-(thiazole-2-carbonyl)-butyl]-amide | CHEMBL320781
Type:
Small organic molecule
Emp. Form.:
C28H35N9O4S
Mol. Mass.:
593.7
SMILES:
N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N1C[C@@H]2CC[C@H](N2C(=O)C1)C(=O)NC(CCCNC(N)=N)C(=O)c1nccs1