Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 1A2
Ligand
BDBM50314108
Substrate
n/a
Meas. Tech.
ChEMBL_627649 (CHEMBL1114479)
IC50
>100000±n/a nM
Citation
 Debenham, JS; Madsen-Duggan, CB; Toupence, RB; Walsh, TF; Wang, J; Tong, X; Kumar, S; Lao, J; Fong, TM; Xiao, JC; Huang, CR; Shen, CP; Feng, Y; Marsh, DJ; Stribling, DS; Shearman, LP; Strack, AM; Goulet, MT Furo[2,3-b]pyridine-based cannabinoid-1 receptor inverse agonists: synthesis and biological evaluation. Part 1. Bioorg Med Chem Lett 20:1448-52 (2010) [PubMed] Article
Debenham, JS; Madsen-Duggan, CB; Toupence, RB; Walsh, TF; Wang, J; Tong, X; Kumar, S; Lao, J; Fong, TM; Xiao, JC; Huang, CR; Shen, CP; Feng, Y; Marsh, DJ; Stribling, DS; Shearman, LP; Strack, AM; Goulet, MT Furo[2,3-b]pyridine-based cannabinoid-1 receptor inverse agonists: synthesis and biological evaluation. Part 1. Bioorg Med Chem Lett 20:1448-52 (2010) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 1A2
Synonyms:
CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3
Type:
Enzyme
Mol. Mass.:
58423.38
Organism:
Homo sapiens (Human)
Description:
P05177
Residue:
516
Sequence:
MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKNPHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDGQSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELMAGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFPILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGNLIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLSDRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPELWEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLEFSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
Inhibitor
Name:
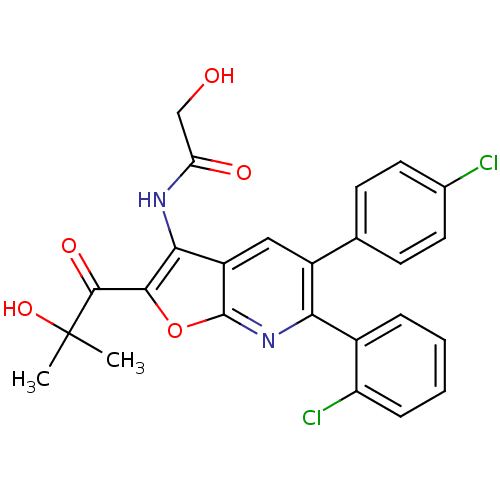
BDBM50314108
Synonyms:
CHEMBL1092169 | N-(6-(2-chlorophenyl)-5-(4-chlorophenyl)-2-(2-hydroxy-2-methylpropanoyl)furo[2,3-b]pyridin-3-yl)-2-hydroxyacetamide
Type:
Small organic molecule
Emp. Form.:
C25H20Cl2N2O5
Mol. Mass.:
499.343
SMILES:
CC(C)(O)C(=O)c1oc2nc(-c3ccccc3Cl)c(cc2c1NC(=O)CO)-c1ccc(Cl)cc1