Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cathepsin D
Ligand
BDBM50281638
Substrate
n/a
Meas. Tech.
ChEBML_45171
Ki
200±n/a nM
Citation
 Sawyer, T; Fisher, J; Hester, J; Smith, C; Tomasselli, A; Tarpley, W; Burton, P; Hui, J; McQuade, T; Conradi, R; Bradford, V; Liu, L; Kinner, J; Tustin, J; Alexander, D; Harrison, A; Emmert, D; Staples, D; Maggiora, L; Zhang, Y; Poorman, R; Dunna, B; Rao, C; Scarborough, P; Lowther, W; Craik, C; DeCamp, D; Moon, J; Howe, W; Heinrikson, R Peptidomimetic inhibitors of human immunodeficiency virus protease (HIV-PR): Design, enzyme binding and selectivity, antiviral efficacy, and cell permeability properties Bioorg Med Chem Lett 3:819-824 (1993) Article
Sawyer, T; Fisher, J; Hester, J; Smith, C; Tomasselli, A; Tarpley, W; Burton, P; Hui, J; McQuade, T; Conradi, R; Bradford, V; Liu, L; Kinner, J; Tustin, J; Alexander, D; Harrison, A; Emmert, D; Staples, D; Maggiora, L; Zhang, Y; Poorman, R; Dunna, B; Rao, C; Scarborough, P; Lowther, W; Craik, C; DeCamp, D; Moon, J; Howe, W; Heinrikson, R Peptidomimetic inhibitors of human immunodeficiency virus protease (HIV-PR): Design, enzyme binding and selectivity, antiviral efficacy, and cell permeability properties Bioorg Med Chem Lett 3:819-824 (1993) Article More Info.:
Target
Name:
Cathepsin D
Synonyms:
CATD_HUMAN | CPSD | CTSD | Cathepsin D [Precursor] | Cathepsin D heavy chain | Cathepsin D light chain | Cathepsin D precursor
Type:
Enzyme
Mol. Mass.:
44551.72
Organism:
Homo sapiens (Human)
Description:
Human proCathepsin D (SwissProt accession number P07339) was expressed in Sf9 cells, purified, and autoactivated.
Residue:
412
Sequence:
MQPSSLLPLALCLLAAPASALVRIPLHKFTSIRRTMSEVGGSVEDLIAKGPVSKYSQAVPAVTEGPIPEVLKNYMDAQYYGEIGIGTPPQCFTVVFDTGSSNLWVPSIHCKLLDIACWIHHKYNSDKSSTYVKNGTSFDIHYGSGSLSGYLSQDTVSVPCQSASSASALGGVKVERQVFGEATKQPGITFIAAKFDGILGMAYPRISVNNVLPVFDNLMQQKLVDQNIFSFYLSRDPDAQPGGELMLGGTDSKYYKGSLSYLNVTRKAYWQVHLDQVEVASGLTLCKEGCEAIVDTGTSLMVGPVDEVRELQKAIGAVPLIQGEYMIPCEKVSTLPAITLKLGGKGYKLSPEDYTLKVSQAGKTLCLSGFMGMDIPPPSGPLWILGDVFIGRYYTVFDRDNNRVGFAEAARL
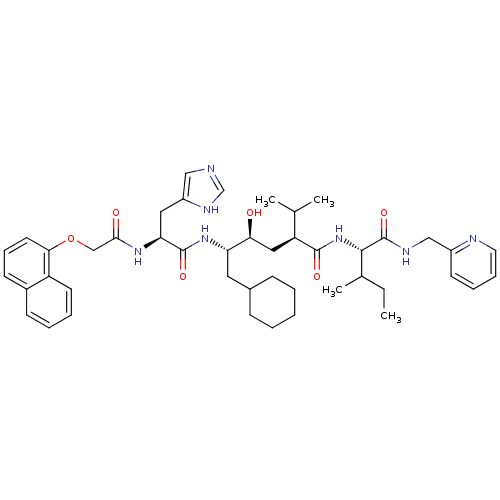
Inhibitor
Name:
BDBM50281638
Synonyms:
(2S,4S,5S)-6-Cyclohexyl-4-hydroxy-5-{(S)-3-(1H-imidazol-4-yl)-2-[2-(naphthalen-1-yloxy)-acetylamino]-propionylamino}-2-isopropyl-hexanoic acid {(S)-2-methyl-1-[(pyridin-2-ylmethyl)-carbamoyl]-butyl}-amide | CHEMBL165706
Type:
Small organic molecule
Emp. Form.:
C45H61N7O6
Mol. Mass.:
796.0091
SMILES:
CCC(C)[C@H](NC(=O)[C@@H](C[C@H](O)[C@H](CC1CCCCC1)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)COc1cccc2ccccc12)C(C)C)C(=O)NCc1ccccn1