Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Tyrosine-protein kinase Lck
Ligand
BDBM50236362
Substrate
n/a
Meas. Tech.
ChEMBL_474929 (CHEMBL933002)
IC50
38±n/a nM
Citation
 Harmange, JC; Weiss, MM; Germain, J; Polverino, AJ; Borg, G; Bready, J; Chen, D; Choquette, D; Coxon, A; DeMelfi, T; DiPietro, L; Doerr, N; Estrada, J; Flynn, J; Graceffa, RF; Harriman, SP; Kaufman, S; La, DS; Long, A; Martin, MW; Neervannan, S; Patel, VF; Potashman, M; Regal, K; Roveto, PM; Schrag, ML; Starnes, C; Tasker, A; Teffera, Y; Wang, L; White, RD; Whittington, DA; Zanon, R Naphthamides as novel and potent vascular endothelial growth factor receptor tyrosine kinase inhibitors: design, synthesis, and evaluation. J Med Chem 51:1649-67 (2008) [PubMed] Article
Harmange, JC; Weiss, MM; Germain, J; Polverino, AJ; Borg, G; Bready, J; Chen, D; Choquette, D; Coxon, A; DeMelfi, T; DiPietro, L; Doerr, N; Estrada, J; Flynn, J; Graceffa, RF; Harriman, SP; Kaufman, S; La, DS; Long, A; Martin, MW; Neervannan, S; Patel, VF; Potashman, M; Regal, K; Roveto, PM; Schrag, ML; Starnes, C; Tasker, A; Teffera, Y; Wang, L; White, RD; Whittington, DA; Zanon, R Naphthamides as novel and potent vascular endothelial growth factor receptor tyrosine kinase inhibitors: design, synthesis, and evaluation. J Med Chem 51:1649-67 (2008) [PubMed] Article More Info.:
Target
Name:
Tyrosine-protein kinase Lck
Synonyms:
2.7.10.2 | LCK | LCK_HUMAN | LSK | Leukocyte C-terminal Src kinase | Lymphocyte cell-specific protein-tyrosine kinase | Lymphocyte-specific protein tyrosine kinase | P56-LCK | Protein YT16 | Proto-oncogene Lck | Proto-oncogene tyrosine-protein kinase LCK | Src/Lck kinase | T cell-specific protein-tyrosine kinase
Type:
n/a
Mol. Mass.:
57987.83
Organism:
Homo sapiens (Human)
Description:
P06239
Residue:
509
Sequence:
MGCGCSSHPEDDWMENIDVCENCHYPIVPLDGKGTLLIRNGSEVRDPLVTYEGSNPPASPLQDNLVIALHSYEPSHDGDLGFEKGEQLRILEQSGEWWKAQSLTTGQEGFIPFNFVAKANSLEPEPWFFKNLSRKDAERQLLAPGNTHGSFLIRESESTAGSFSLSVRDFDQNQGEVVKHYKIRNLDNGGFYISPRITFPGLHELVRHYTNASDGLCTRLSRPCQTQKPQKPWWEDEWEVPRETLKLVERLGAGQFGEVWMGYYNGHTKVAVKSLKQGSMSPDAFLAEANLMKQLQHQRLVRLYAVVTQEPIYIITEYMENGSLVDFLKTPSGIKLTINKLLDMAAQIAEGMAFIEERNYIHRDLRAANILVSDTLSCKIADFGLARLIEDNEYTAREGAKFPIKWTAPEAINYGTFTIKSDVWSFGILLTEIVTHGRIPYPGMTNPEVIQNLERGYRMVRPDNCPEELYQLMRLCWKERPEDRPTFDYLRSVLEDFFTATEGQYQPQP
Inhibitor
Name:
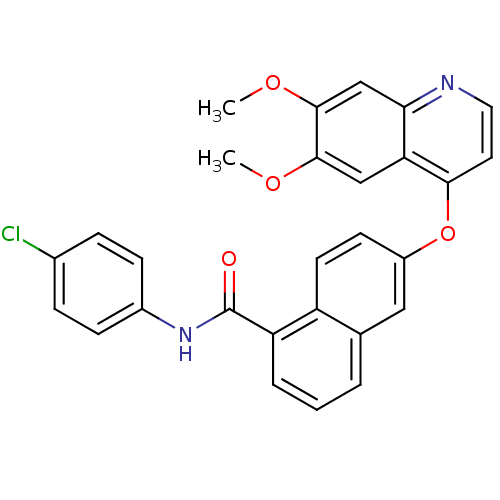
BDBM50236362
Synonyms:
CHEMBL429743 | N-(4-chlorophenyl)-6-(6,7-dimethoxyquinolin-4-yloxy)-1-naphthamide | N-(4-chlorophenyl)-6-[(6,7-dimethoxyquinolin-4-yl)oxy]naphthalene-1-carboxamide
Type:
Small organic molecule
Emp. Form.:
C28H21ClN2O4
Mol. Mass.:
484.93
SMILES:
COc1cc2nccc(Oc3ccc4c(cccc4c3)C(=O)Nc3ccc(Cl)cc3)c2cc1OC