Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Neuraminidase
Ligand
BDBM5265
Substrate
BDBM4702
Meas. Tech.
Neuraminidase Inhibition Assay
pH
6.5±n/a
Temperature
310.15±n/a K
IC50
4±n/a nM
Citation
 Atigadda, VR; Brouillette, WJ; Duarte, F; Babu, YS; Bantia, S; Chand, P; Chu, N; Montgomery, JA; Walsh, DA; Sudbeck, E; Finley, J; Air, GM; Luo, M; Laver, GW Hydrophobic benzoic acids as inhibitors of influenza neuraminidase. Bioorg Med Chem 7:2487-97 (1999) [PubMed] Article
Atigadda, VR; Brouillette, WJ; Duarte, F; Babu, YS; Bantia, S; Chand, P; Chu, N; Montgomery, JA; Walsh, DA; Sudbeck, E; Finley, J; Air, GM; Luo, M; Laver, GW Hydrophobic benzoic acids as inhibitors of influenza neuraminidase. Bioorg Med Chem 7:2487-97 (1999) [PubMed] Article More Info.:
Target
Name:
Neuraminidase
Synonyms:
Influenza B Virus Neuraminidase | NA | NRAM_INBLE | Neuraminidase | Neuraminidase B
Type:
Enzyme
Mol. Mass.:
51446.67
Organism:
Influenza B virus (B/Lee/40)
Description:
n/a
Residue:
466
Sequence:
MLPSTVQTLTLLLTSGGVLLSLYVSASLSYLLYSDVLLKFSSTKTTAPTMSLECTNASNAQTVNHSATKEMTFPPPEPEWTYPRLSCQGSTFQKALLISPHRFGEIKGNSAPLIIREPFVACGPKECRHFALTHYAAQPGGYYNGTRKDRNKLRHLVSVKLGKIPTVENSIFHMAAWSGSACHDGREWTYIGVDGPDNDALVKIKYGEAYTDTYHSYAHNILRTQESACNCIGGDCYLMITDGSASGISKCRFLKIREGRIIKEILPTGRVEHTEECTCGFASNKTIECACRDNSYTAKRPFVKLNVETDTAEIRLMCTKTYLDTPRPDDGSIAGPCESNGDKWLGGIKGGFVHQRMASKIGRWYSRTMSKTNRMGMELYVKYDGDPWTDSDALTLSGVMVSIEEPGWYSFGFEIKDKKCDVPCIGIEMVHDGGKDTWHSAATAIYCLMGSGQLLWDTVTGVDMAL
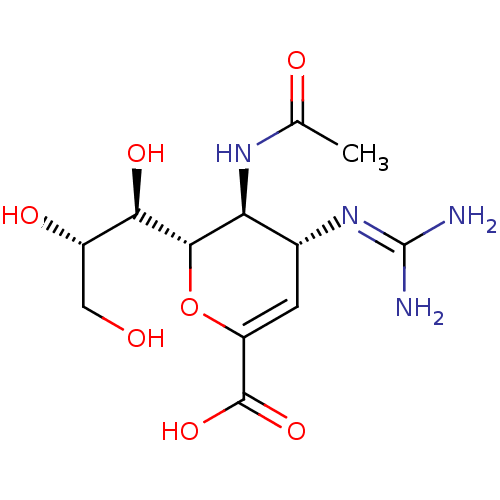
Inhibitor
Name:
BDBM5265
Synonyms:
(2S,3S,4R)-4-carbamimidamido-3-acetamido-2-[(1R,2S)-1,2,3-trihydroxypropyl]-3,4-dihydro-2H-pyran-6-carboxylic acid | Benzoic Acid Inhibitor 3
Type:
Small organic molecule
Emp. Form.:
C12H20N4O7
Mol. Mass.:
332.3098
SMILES:
[#6]-[#6](=O)-[#7]-[#6@H]-1-[#6@@H](-[#6]=[#6](-[#8]-[#6@@H]-1-[#6@H](-[#8])-[#6@@H](-[#8])-[#6]-[#8])-[#6](-[#8])=O)\[#7]=[#6](/[#7])-[#7] |r,c:6|
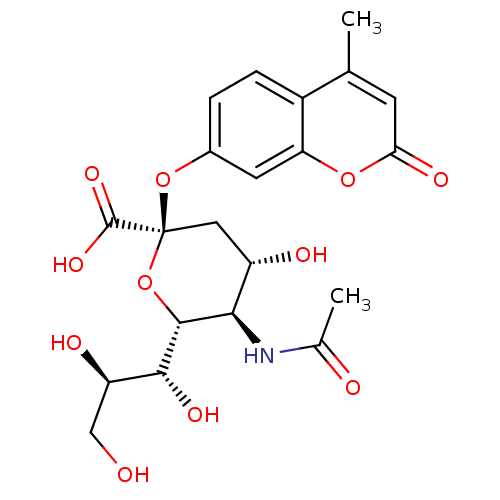
Substrate
Name:
BDBM4702
Synonyms:
(2R,4S,5R,6R)-5-acetamido-4-hydroxy-2-[(4-methyl-2-oxo-2H-chromen-7-yl)oxy]-6-[(1R,2R)-1,2,3-trihydroxypropyl]oxane-2-carboxylic acid | 2 -(4-methylumbelliferyl)-alpha-D-acetylneuraminic acid | neuraminidase substrate
Type:
Small organic molecule
Emp. Form.:
C21H25NO11
Mol. Mass.:
467.4233
SMILES:
CC(=O)N[C@@H]1[C@@H](O)C[C@](Oc2ccc3c(C)cc(=O)oc3c2)(O[C@H]1[C@H](O)[C@H](O)CO)C(O)=O |r|