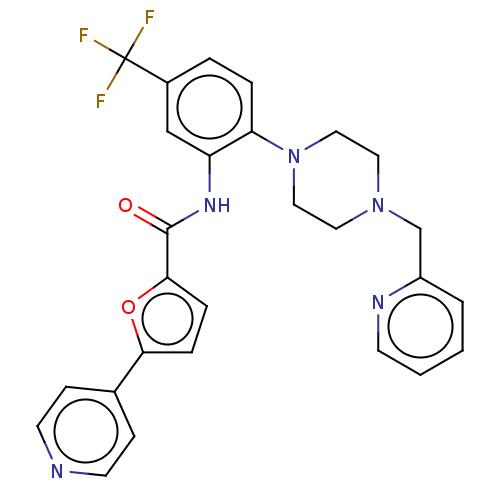
BDBM221074 SPHINX31::US9695160, 12::US9796707, Compound 12
SMILES FC(F)(F)c1ccc(N2CCN(Cc3ccccn3)CC2)c(NC(=O)c2ccc(o2)-c2ccncc2)c1
InChI Key InChIKey=VURLRACCOCGFDB-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 221074
Found 6 hits for monomerid = 221074
Affinity DataIC50: 6nMpH: 7.5 T: 2°CAssay Description:A reaction buffer containing 200 mM Tris pH 7.5, 100 mM MgCl2 and 0.1 mg/ml BSA was added to 43 μM SRSF1Arg-Ser (RS) peptide (NH2-RSPSYGRSRSRSRS...More data for this Ligand-Target Pair
Affinity DataIC50: 5.90nMpH: 7.5 T: 2°CAssay Description:A reaction buffer containing 200 mM Tris pH 7.5, 100 mM MgCl2 and 0.1 mg/ml BSA was added to 43 μM SRSF1Arg-Ser (RS) peptide (NH2-RSPSYGRSRSRSRS...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Inhibition of SRPK1 (unknown origin) using SRSF1 Arg-Ser (RS) peptide (NH2-RSPSYGRSRSRSRSRSRSRSRSNSRSRSY-OH) as substrate incubated for 10 mins in pr...More data for this Ligand-Target Pair
Affinity DataIC50: 3.18nMT: 2°CAssay Description:Candidate compounds were screened by the Kinase-Glo assay (Promega; Koresawa and Okabe, 2004), the results of which are shown in Table 1 and Table 2....More data for this Ligand-Target Pair
Affinity DataIC50: 3.18nMAssay Description:Candidate compounds were screened by the Kinase-Glo assay (Promega; Koresawa and Okabe, 2004), the results of which are shown in Table 1 and Table 2....More data for this Ligand-Target Pair
Affinity DataIC50: 300nMpH: 7.45 T: 2°CAssay Description:Whole cell patch clamp recordings of hERG current (IhERG) were made at 37 °C from human embryonic kidney cells (HEK 293) cells stably expressing ...More data for this Ligand-Target Pair
Activity Spreadsheet -- ITC Data from BindingDB
 Found 1 hit for monomerid = 221074
Found 1 hit for monomerid = 221074
ITC DataΔG°: -9.90kcal/mole −TΔS°: -3.84kcal/mole ΔH°: -5.97kcal/mole logk: 1.39E+7
pH: 7.5 T: 30.00°C
pH: 7.5 T: 30.00°C
 3D Structure (crystal)
3D Structure (crystal)