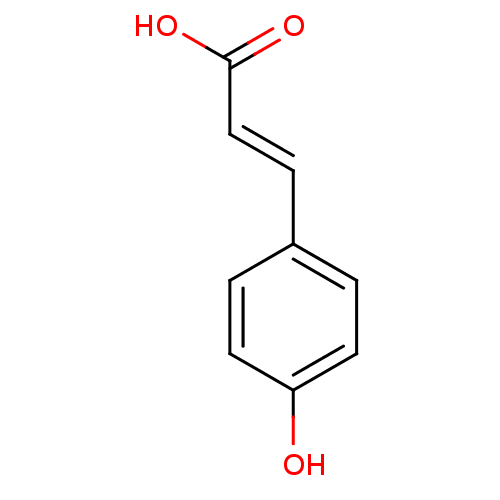
BDBM4374 (2E)-3-(4-hydroxyphenyl)prop-2-enoic acid::(2E)-3-(4-hydroxyphenyl)prop-2-enoic acid (8)::CHEMBL66879::p-Coumaric Acid::p-Hydroxycinnamic acid (p-HCA)::p-hydroxycinnamic acid::p-hydroxycinnamic acid (M4)
SMILES OC(=O)\C=C\c1ccc(O)cc1
InChI Key InChIKey=NGSWKAQJJWESNS-ZZXKWVIFSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 38 hits for monomerid = 4374
Found 38 hits for monomerid = 4374
Affinity DataKi: 600nM ΔG°: -8.48kcal/molepH: 7.5 T: 2°CAssay Description:An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity [Khalifah et al., J. Biol. Chem., ...More data for this Ligand-Target Pair
Affinity DataKi: 980nMAssay Description:Inhibition of human CA2 by stopped-flow CO2 hydration assayMore data for this Ligand-Target Pair
Affinity DataKi: 1.07E+3nMAssay Description:Inhibition of human CA1 by stopped-flow CO2 hydration assayMore data for this Ligand-Target Pair
Affinity DataKi: 1.56E+3nM ΔG°: -7.92kcal/molepH: 7.5 T: 2°CAssay Description:An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity [Khalifah et al., J. Biol. Chem., ...More data for this Ligand-Target Pair
Affinity DataKi: 3.03E+3nM ΔG°: -7.52kcal/molepH: 7.5 T: 2°CAssay Description:An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity [Khalifah et al., J. Biol. Chem., ...More data for this Ligand-Target Pair
Affinity DataKi: 5.23E+3nMAssay Description:Inhibition of human CA7 by stopped-flow CO2 hydration assayMore data for this Ligand-Target Pair
Affinity DataKi: 5.33E+3nMAssay Description:Inhibition of human CA9 by stopped-flow CO2 hydration assayMore data for this Ligand-Target Pair
Affinity DataKi: 5.96E+3nMAssay Description:Inhibition of human CA5A by stopped-flow CO2 hydration assayMore data for this Ligand-Target Pair
Affinity DataKi: 6.68E+3nMAssay Description:Inhibition of human CA14 by stopped-flow CO2 hydration assayMore data for this Ligand-Target Pair
Affinity DataKi: 6.68E+3nMAssay Description:Inhibition of human carbonic anhydrase 14 preincubated for 15 mins by stopped-flow CO2 hydration assayMore data for this Ligand-Target Pair
Affinity DataKi: 6.72E+3nMAssay Description:Inhibition of human CA6 by stopped-flow CO2 hydration assayMore data for this Ligand-Target Pair
Affinity DataKi: 7.57E+3nMAssay Description:Inhibition of human CA3 by stopped-flow CO2 hydration assayMore data for this Ligand-Target Pair
Affinity DataKi: 7.76E+3nMAssay Description:Inhibition of human CA5B by stopped-flow CO2 hydration assayMore data for this Ligand-Target Pair
Affinity DataKi: 8.01E+3nMAssay Description:Inhibition of human catalytic domain of carbonic anhydrase 12 preincubated for 15 mins by stopped-flow CO2 hydration assayMore data for this Ligand-Target Pair
Affinity DataKi: 8.01E+3nMAssay Description:Inhibition of human CA12 by stopped-flow CO2 hydration assayMore data for this Ligand-Target Pair
Affinity DataKi: 9.60E+3nMAssay Description:Inhibition of human CA4 by stopped-flow CO2 hydration assayMore data for this Ligand-Target Pair
Affinity DataKi: 1.01E+4nMAssay Description:Inhibition of mouse carbonic anhydrase 13 preincubated for 15 mins by stopped-flow CO2 hydration assayMore data for this Ligand-Target Pair
Affinity DataKi: 1.01E+4nMAssay Description:Inhibition of mouse CA13 by stopped-flow CO2 hydration assayMore data for this Ligand-Target Pair
Affinity DataKi: 1.40E+4nMAssay Description:Displacement of [3H]nicotinic acid from human GPR109a receptor expressed in human HEK293T cells by liquid scintillation countingMore data for this Ligand-Target Pair
TargetCalcium/calmodulin-dependent protein kinase type II subunit alpha(Homo sapiens (Human))
University Of Copenhagen
Curated by ChEMBL
University Of Copenhagen
Curated by ChEMBL
Affinity DataKi: >1.00E+5nMAssay Description:Inhibition of [3H]NCS-382 binding to CaMK2alpha (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 4.41E+5nM ΔG°: -4.57kcal/molepH: 7.4 T: 2°CAssay Description:Carbonic anhydrase activity was assayed by following the change in absorbance at 348 nm of 4-nitrophenylacetate (NPA) to 4-nitrophenylate ion using s...More data for this Ligand-Target Pair
Affinity DataKi: 5.37E+5nM ΔG°: -4.46kcal/molepH: 7.4 T: 2°CAssay Description:Carbonic anhydrase activity was assayed by following the change in absorbance at 348 nm of 4-nitrophenylacetate (NPA) to 4-nitrophenylate ion using s...More data for this Ligand-Target Pair
Affinity DataKi: 1.00E+6nM IC50: 3.00E+6nMAssay Description:The activity of EGFR, preactivated with EGF, is measured by its ability to transfer terminal phosphate from [gamma-32P]ATP to poly(GAT) substrate.More data for this Ligand-Target Pair
TargetTyrosine-protein phosphatase non-receptor type 1 [1-321](Homo sapiens (Human))
Chulalongkorn University
Chulalongkorn University
Affinity DataIC50: 1.68E+5nMpH: 7.5 T: 2°CAssay Description:PTP1B was diluted before the experiment to 1.2 μg/mL in Tris buffer, pH7.6 (10 mM Tris, 1.0 mM EDTA, 3.0 mM DTT, 0.01% w/v NaN3). The tested com...More data for this Ligand-Target Pair
Affinity DataIC50: 9.08E+4nMAssay Description:The assay was performed using isolated phenolics from maize, and inhibition was determined according to previously described method.More data for this Ligand-Target Pair
Affinity DataIC50: 6.80E+3nMpH: 7.6 T: 2°CAssay Description:Inhibition of xanthine oxidase (XO) by each isolated phenolics was measured by following the decrease in the uric acid formation at 293nm at 25°...More data for this Ligand-Target Pair
Affinity DataIC50: 140nMAssay Description:Inhibition of aldose reductase (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: >3.00E+5nMAssay Description:Inhibition of yeast alpha-glucosidase using p-nitrophenyl-alpha-D-glucopyranoside as substrate preincubated for 10 min before substrate addition and ...More data for this Ligand-Target Pair
TargetEstrogen receptor(Homo sapiens (Human))
East China University Of Science And Technology
Curated by ChEMBL
East China University Of Science And Technology
Curated by ChEMBL
Affinity DataEC50: 1.61E+4nMAssay Description:Agonist activity at human estrogen receptor-alpha by yeast two-hybrid assay in presence of SRC1More data for this Ligand-Target Pair
TargetEstrogen receptor beta(Homo sapiens (Human))
East China University Of Science And Technology
Curated by ChEMBL
East China University Of Science And Technology
Curated by ChEMBL
Affinity DataEC50: 4.89E+3nMAssay Description:Agonist activity at human estrogen receptor-beta by yeast two-hybrid assay in presence of SRC1More data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+11nMAssay Description:Inhibition of baker's yeast alpha-glucosidase using p-nitrophenyl-alpha-D-glucopyranoside as substrate by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of human tyrosinase after 120 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of human tyrosinase after 120 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+3nMAssay Description:Inhibition of tyrosinase in neonatal human epidermal melanocytesMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+3nMAssay Description:Inhibition of human tyrosinase expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 7.60E+4nMAssay Description:Inhibition of human recombinant N-terminus His6-tagged AKR1B1 expressed in Escherichia coli BL21 DE3 assessed as pyridine-3-aldehyde reduction by spe...More data for this Ligand-Target Pair
Affinity DataIC50: 1.21E+5nMAssay Description:Inhibition of mushroom tyrosinase after 25 mins by spectrophotometryMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B10(Homo sapiens (Human))
Gifu Pharmaceutical University
Curated by ChEMBL
Gifu Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 6.30E+4nMAssay Description:Inhibition of human recombinant N-terminus His6-tagged AKR1B10 expressed in Escherichia coli BL21 DE3 assessed as pyridine-3-aldehyde reduction by sp...More data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)