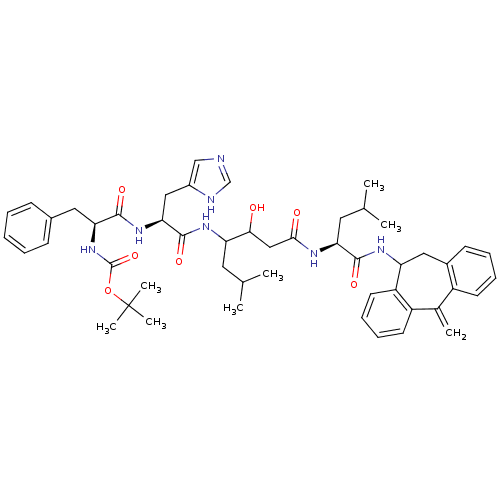
BDBM50023093 Boc-Phe-His-Sta-Leu-10,11-dihydro-5H-dibenzo[a,d]-cycloheptenamide::CHEMBL297880
SMILES CC(C)CC(NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](Cc1ccccc1)NC(=O)OC(C)(C)C)C(O)CC(=O)N[C@@H](CC(C)C)C(=O)NC1Cc2ccccc2C(=C)c2ccccc12
InChI Key InChIKey=JPVWOKQLLOKBLS-PWNZBVGMSA-N
Data 3 KI
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50023093
Found 3 hits for monomerid = 50023093
Affinity DataKi: 120nMAssay Description:Inhibition of purified human kidney renin, fluorometric assay using a synthetic tetradecapeptide renin substrate at 10e-9 M concentrationMore data for this Ligand-Target Pair
Affinity DataKi: 320nMAssay Description:Inhibition of purified human kidney renin, fluorometric assay using a synthetic tetradecapeptide renin substrate at 10e-9 M concentrationMore data for this Ligand-Target Pair
Affinity DataKi: 520nMAssay Description:Inhibition of purified human kidney renin, radioimmunoassay using a synthetic tetradecapeptide renin substrate at 10e-9 M concentrationMore data for this Ligand-Target Pair