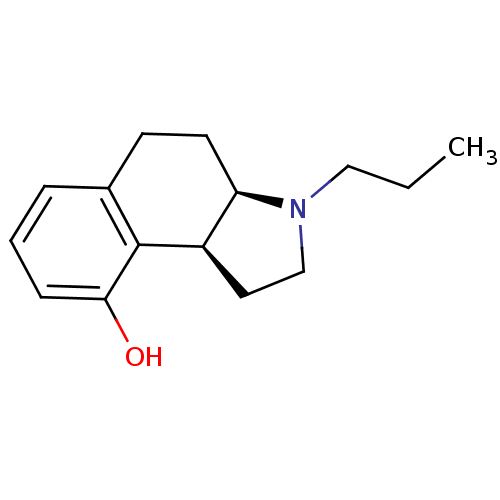
BDBM50034342 (3aR,9bS) 3-Propyl-2,3,3a,4,5,9b-hexahydro-1H-benzo[e]indol-9-ol::(3aR,9bS)-3-Propyl-2,3,3a,4,5,9b-hexahydro-1H-benzo[e]indol-9-ol::(3aS,9bR) 3-Propyl-2,3,3a,4,5,9b-hexahydro-1H-benzo[e]indol-9-ol::(Recemic)3-Propyl-2,3,3a,4,5,9b-hexahydro-1H-benzo[e]indol-9-ol::CHEMBL430332
SMILES CCCN1CC[C@@H]2[C@H]1CCc1cccc(O)c21
InChI Key InChIKey=UVOXCNCVSFLEPX-CHWSQXEVSA-N
Data 11 KI
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 11 hits for monomerid = 50034342
Found 11 hits for monomerid = 50034342
Affinity DataKi: 0.100nMAssay Description:Binding affinity of the compound against 5-hydroxytryptamine 1A receptorMore data for this Ligand-Target Pair
Affinity DataKi: 0.200nMAssay Description:Tested in vitro for the inhibition of [3H]-8-OH-DPAT binding to 5-hydroxytryptamine 1A receptor, expressed in cloned CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Binding affinity against 5-hydroxytryptamine 1A receptor from bovine hippocampus, used [3H]8-OH-DPAT as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Binding affinity against 5-hydroxytryptamine 1A receptor from bovine hippocampus, used [3H]8-OH-DPAT as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 3nMAssay Description:Binding affinity against 5-hydroxytryptamine 1A receptor from bovine hippocampus, used [3H]8-OH-DPAT as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 3nMAssay Description:Tested for activity against 5-hydroxytryptamine 1A receptor from bovine hippocampusMore data for this Ligand-Target Pair
Affinity DataKi: <50nMAssay Description:Binding affinity against 5-hydroxytryptamine 1A receptor from bovine hippocampus, used [3H]8-OH-DPAT as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 769nMAssay Description:Tested in vitro for the inhibition of [3H]-raclopride binding to Dopamine receptor D2 in rat striatumMore data for this Ligand-Target Pair
Affinity DataKi: 1.64E+3nMAssay Description:Tested in vitro for the inhibition of [3H]ketanserin binding to 5-hydroxytryptamine 2 receptor, expressed in cloned CHO cells.More data for this Ligand-Target Pair
Affinity DataKi: >4.07E+3nMAssay Description:Tested in vitro for the inhibition of [3H]-spiperone binding to Dopamine receptor D4, expressed in cloned CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: >4.76E+3nMAssay Description:Tested in vitro for the inhibition of [3H]-U-86,170 binding to Dopamine receptor D3, expressed in cloned CHO cellsMore data for this Ligand-Target Pair