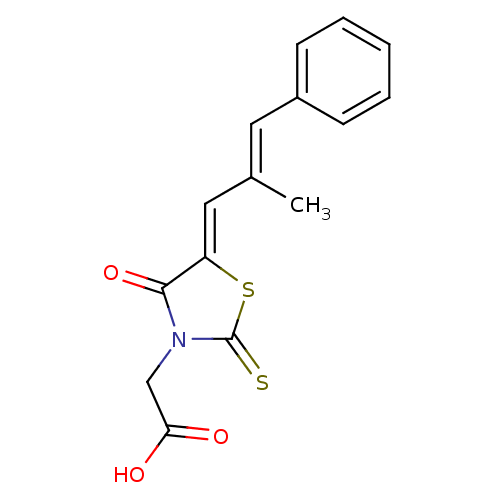
BDBM50049730 2-(5-(2-methyl-3-phenylallylidene)-4-oxo-2-thioxothiazolidin-3-yl)acetic acid::CHEMBL56337::Epalrestat::{5-[(E)-2-Methyl-3-phenyl-prop-2-en-(Z)-ylidene]-4-oxo-2-thioxo-thiazolidin-3-yl}-acetic acid
SMILES C\C(\C=C1/SC(=S)N(CC(O)=O)C1=O)=C/c1ccccc1
InChI Key InChIKey=CHNUOJQWGUIOLD-NFZZJPOKSA-N
Data 61 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 61 hits for monomerid = 50049730
Found 61 hits for monomerid = 50049730
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 1.50E+3nMAssay Description:Invitro inhibitory activity of the compound against rat kidney Aldehyde reductaseMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 45nMAssay Description:Inhibition of rat lens ALR2 using D,L-glyceraldehyde as substrate preincubated for 5 mins at 30 degC followed by substrate addition and measured afte...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 21nMAssay Description:Inhibitory activity against rat lens aldose reductase(AR).More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member A1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 1.50E+3nMAssay Description:In vitro inhibitory activity against rat kidney aldehyde reductase(ALR).More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1 [K65Q](Bos taurus (Cattle))
University Of Messina
Curated by ChEMBL
University Of Messina
Curated by ChEMBL
Affinity DataIC50: 170nMAssay Description:Inhibition of bovine aldose reductase assessed as oxidation of NADPHMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B10(Homo sapiens (Human))
Gifu Pharmaceutical University
Curated by ChEMBL
Gifu Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 330nMAssay Description:Inhibition of reductase activity of N-terminal 6His-tagged AKR1B10 expressed in Escherichia coli BL21(DE3) assessed as inhibition of NADPH linked pyr...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B10(Homo sapiens (Human))
Gifu Pharmaceutical University
Curated by ChEMBL
Gifu Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 330nMAssay Description:Inhibition of recombinant N-His6-tagged AKR1B10 expressed in Escherichia coli BL21 cells using pyridine-3-aldehyde as substrate by spectrophotometryMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 72nMAssay Description:Inhibition of aldose reductase in rat lens homogenates by fluorophotometerMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 250nMAssay Description:Inhibition of Wistar rat lens aldose reductase using D,L-glyceraldehyde as substrate incubated for 1 min measured for 4 mins by spectrophotometryMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 130nMAssay Description:Inhibition of ALR2 from rat lens using D,L-glyceraldehyde as substrate measured as absorption of NADPH for 4 mins by UV/vis spectrophotmetryMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Homo sapiens (Human))
Gifu Pharmaceutical University
Curated by ChEMBL
Gifu Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 21nMAssay Description:Inhibition of N-terminal 6His-tagged human aldose reductase expressed in Escherichia coli BL21(DE3) mediated NADPH linked pyridine-3-aldehyde reducti...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member A1(Homo sapiens (Human))
Gifu Pharmaceutical University
Curated by ChEMBL
Gifu Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 2.60E+3nMAssay Description:Inhibition of N-terminal 6His-tagged human aldehyde reductase expressed in Escherichia coli BL21(DE3) mediated D-glucuronate reductionMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Homo sapiens (Human))
Gifu Pharmaceutical University
Curated by ChEMBL
Gifu Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 1.25E+4nMAssay Description:Inhibition human recombinant aldose reductase 1 by spectrophotometric analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 6.77E+4nMAssay Description:Inhibition sorbitol dehydrogenase by spectrophotometric analysisMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1 [K65Q](Bos taurus (Cattle))
University Of Messina
Curated by ChEMBL
University Of Messina
Curated by ChEMBL
Affinity DataIC50: 170nMAssay Description:Inhibition of bovine lens aldose reductase assessed as inhibition of NDAPH oxidation by non-linear regression analysisMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1 [K65Q](Bos taurus (Cattle))
University Of Messina
Curated by ChEMBL
University Of Messina
Curated by ChEMBL
Affinity DataIC50: 170nMAssay Description:Inhibition of bovine lens ALR2More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 72nMAssay Description:Inhibition of rat lens aldose reductase using DL-glyceraldehyde as substrate after 30 mins by fluorescence microplate reader analysisMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Homo sapiens (Human))
Gifu Pharmaceutical University
Curated by ChEMBL
Gifu Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:Inhibition of human recombinant AKR1B1 expressed in Escherichia coli BL21 cells using pyridine-3-aldehyde as substrate by spectrophotometryMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member A1(Homo sapiens (Human))
Gifu Pharmaceutical University
Curated by ChEMBL
Gifu Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 2.60E+3nMAssay Description:Inhibition of human recombinant AKR1A1 expressed in Escherichia coli BL21 cells using D-glucuronate as substrate by spectrophotometryMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of crude aldose reductase of rat lensMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 13nMAssay Description:Inhibitory activity measured against rat lens aldose reductase using 3-pyridinecarboxaldehyde as substrateChecked by AuthorMore data for this Ligand-Target Pair
Affinity DataIC50: 1.50E+3nMAssay Description:Selectivity ratio measured as the IC50 ratio of aldehyde/aldose reductase valuesChecked by AuthorMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 23nMAssay Description:Inhibitory activity against purified rat lens aldose reductase (RLAR)More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Homo sapiens (Human))
Gifu Pharmaceutical University
Curated by ChEMBL
Gifu Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibitory activity against aldose reductase enzymeMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:Inhibition of rat kidney NADPH-dependent aldose reductase assessed as DL-glyceraldehyde conversion to glycerol preincubated for 20 mins followed by N...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 70nMAssay Description:Inhibition of rat lens aldose reductaseMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 67nMAssay Description:Inhibition of Sprague-Dawley rat lens aldose reductaseMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Homo sapiens (Human))
Gifu Pharmaceutical University
Curated by ChEMBL
Gifu Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 85nMAssay Description:Inhibition of recombinant human ALR2 assessed as reduction in NADPH oxidation measured for 5 mins in presence of D,L-glyceraldehyde by spectrophotome...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 86nMAssay Description:Inhibitory concentration against human erythrocytic purine nucleoside phosphorylase in the presence of 50 mM (pi) orthophosphonateMore data for this Ligand-Target Pair
Affinity DataIC50: 8.44E+4nMAssay Description:Inhibition of human MRP2 overexpressed in Sf9 cell membrane vesicles assessed as uptake of [3H]-estradiol-17beta-D-glucuronide in presence of ATP and...More data for this Ligand-Target Pair
Affinity DataIC50: 6.50E+3nMAssay Description:Inhibition of human MRP4 overexpressed in Sf9 cell membrane vesicles assessed as uptake of [3H]-estradiol-17beta-D-glucuronide in presence of ATP and...More data for this Ligand-Target Pair
Affinity DataIC50: 3.68E+4nMAssay Description:Inhibition of human BSEP overexpressed in Sf9 cell membrane vesicles assessed as uptake of [3H]-taurocholate in presence of ATP measured after 15 to ...More data for this Ligand-Target Pair
Affinity DataIC50: 4.58E+4nMAssay Description:Inhibition of human MRP3 overexpressed in Sf9 insect cell membrane vesicles assessed as uptake of [3H]-estradiol-17beta-D-glucuronide in presence of ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Homo sapiens (Human))
Gifu Pharmaceutical University
Curated by ChEMBL
Gifu Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 94nMAssay Description:Inhibition of aldose reductase (unknown origin)More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 227nMAssay Description:Inhibition of ALR2 in Wistar rat eye lens assessed as reduction in NADPH consumption preincubated for 1 min followed by D,L-glyceraldehyde addition m...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 84nMAssay Description:Inhibition of Wistar rat lenses ALR2 using D,L-glyceraldehyde as substrate preincubated for 10 mins before substrate addition measured after 5 mins b...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 120nMAssay Description:Inhibition of Wistar rat eye lens aldose reductase-2 using D-L glyceraldehyde as substrate assessed as oxidation of NADPH preincubated for 10 mins fo...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 86nMAssay Description:Inhibition of Wistar rat ALR2 using D,L-glyceraldehyde as substrate assessed as oxidation of NADPH preincubated for 10 mins followed by substrate add...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Homo sapiens (Human))
Gifu Pharmaceutical University
Curated by ChEMBL
Gifu Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 93.9nMAssay Description:Inhibition of human muscle recombinant aldose reductase by spectrophotometryMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 170nMAssay Description:Inhibition of Sprague-Dawley albino rat ALR2 assessed as enzyme-mediated oxidation of NADPH using D,L-glyceraldehyde as substrate by spectrophotometr...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 170nMAssay Description:Inhibition of Sprague-Dawley albino rat lens ALR2 by spectrophotometryMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Homo sapiens (Human))
Gifu Pharmaceutical University
Curated by ChEMBL
Gifu Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 620nMAssay Description:Inhibition of human recombinant ALR2 expressed in Escherichia coli using DL-glyceraldehyde as substrate and NADPH preincubated for 5 mins followed by...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 510nMAssay Description:Inhibition of ALR2 in rat erythrocytes assessed as reduction in sorbitol accumulation incubated for 3 hrs in presence of 28 mM glucose by gas chromat...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 860nMAssay Description:Inhibition of ALR2 in rat sciatic nerve assessed as reduction in sorbitol accumulation incubated for 3 hrs in presence of 28 mM glucose by gas chroma...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 530nMAssay Description:Inhibition of ALR2 in rat lens assessed as reduction in sorbitol accumulation incubated for 3 hrs in presence of 28 mM glucose by gas chromatographic...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 3.21E+3nMAssay Description:Inhibition of rat kidney ALR1 using DL-glyceraldehyde as substrate and NADPH preincubated for 5 mins followed by substrate addition and measured for ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Homo sapiens (Human))
Gifu Pharmaceutical University
Curated by ChEMBL
Gifu Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 102nMAssay Description:Inhibition of human recombinant aldose reductase expressed in Escherichia coli BL21 (DE3) pLysS assessed as reduction in NADPH oxidation using L-idos...More data for this Ligand-Target Pair
TargetEnoyl-[acyl-carrier-protein] reductase [NADH](Mycobacterium tuberculosis (strain ATCC 25618 / H3...)
Nanjing University
Curated by ChEMBL
Nanjing University
Curated by ChEMBL
Affinity DataIC50: >2.00E+5nMAssay Description:Inhibition of recombinant Mycobacterium tuberculosis InhA expressed in Escherichia coli Rosette(DE3) pLysS using trans-2-decenoyl-N-acetylcysteamine ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 2.26E+3nMAssay Description:Inhibition of Wistar rat kidney ALR1 using sodium D-glucuronate as substrate preincubated for 10 mins followed by substrate addition and measured for...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member B1(Rattus norvegicus)
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Nippon Zoki Pharmaceutical
Curated by ChEMBL
Affinity DataIC50: 31nMAssay Description:Inhibition of Wistar rat lens ALR2 using L-glyceraldehyde as substrate preincubated for 10 mins followed by substrate addition and measured for 4 min...More data for this Ligand-Target Pair