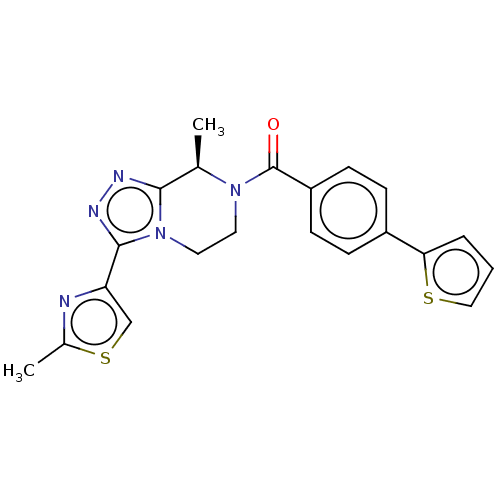
BDBM50081195 CHEMBL3422010::US10065961, Compound 1::US10683295, Compound 1::US10941151, Compound 1::US9475814, 1
SMILES C[C@H]1N(CCn2c1nnc2-c1csc(C)n1)C(=O)c1ccc(cc1)-c1cccs1
InChI Key InChIKey=BWTVMVHFJRFVFO-CYBMUJFWSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 42 hits for monomerid = 50081195
Found 42 hits for monomerid = 50081195
Affinity DataKi: 13nMAssay Description:Displacement of [3H]-SB222200 from recombinant human NK3R expressed in CHO cell membranes after 90 mins by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:Binding affinity to human recombinant NK3R by radioligand binding assayMore data for this Ligand-Target Pair
Affinity DataKi: 16nMAssay Description:NK3: The ability of compounds of the invention to inhibit the binding of the NK-3 receptor selective antagonist 3H-SB222200 was assessed by an in vit...More data for this Ligand-Target Pair
Affinity DataKi: 16nMpH: 7.4Assay Description:The ability of compounds of the invention to inhibit the binding of the NK-3 receptor selective antagonist 3H-SB222200 was assessed by an in vitro ra...More data for this Ligand-Target Pair
Affinity DataKi: 16nMAssay Description:NK3: The ability of compounds of the invention to inhibit the binding of the NK-3 receptor selective antagonist 3H-SB222200 was assessed by an in vit...More data for this Ligand-Target Pair
Affinity DataKi: 16nMAssay Description:NK3: The ability of compounds of the invention to inhibit the binding of the NK-3 receptor selective antagonist 3H-SB222200 was assessed by an in vit...More data for this Ligand-Target Pair
Affinity DataKi: 16nMAssay Description:NK3: The ability of compounds of the invention to inhibit the binding of the NK-3 receptor selective antagonist 3H-SB222200 was assessed by an in vit...More data for this Ligand-Target Pair
Affinity DataKi: 16nMAssay Description:The antagonist activity of compounds of the invention is measured following pre-incubation (3 minutes) of the compound with the cells, followed by ad...More data for this Ligand-Target Pair
Affinity DataKi: 16nMpH: 7.4Assay Description:The ability of compounds of the invention to inhibit the binding of the NK-3 receptor selective antagonist 3H-SB222200 was assessed by an in vitro ra...More data for this Ligand-Target Pair
Affinity DataKi: 20nMAssay Description:Displacement of [3H]-SB222200 from monkey NK3R after 90 mins by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 76nMAssay Description:Displacement of [3H]-SB222200 from rat NK3R after 90 mins by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 76nMAssay Description:Binding affinity to rat NK3RMore data for this Ligand-Target Pair
Affinity DataKi: 79nMAssay Description:Displacement of [3H]-SB222200 from rat NK3R after 90 mins by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 1.26E+4nMAssay Description:Displacement of [3H]-Substance P from human NK1R after 90 mins by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 1.26E+4nMAssay Description:Displacement of [125I]-neurokinin A from human NK2R after 90 mins by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 1.27E+4nM ΔG°: -6.67kcal/molepH: 7.4 T: 2°CAssay Description:The affinity of compounds of the invention for the NK1 receptor was evaluated in CHO recombinant cells which express the human NK1 receptor. Membrane...More data for this Ligand-Target Pair
Affinity DataKi: 1.27E+4nM ΔG°: -6.67kcal/molepH: 7.4 T: 2°CAssay Description:The affinity of compounds of the invention for the NK1 receptor was evaluated in CHO recombinant cells which express the human NK1 receptor. Membrane...More data for this Ligand-Target Pair
Affinity DataKi: 1.27E+4nMAssay Description:NK1: The following radioligand: [3H] substance P (PerkinElmer Cat#NET111520) was used in this assay. Binding assays were performed in a 50 mM Tris/5 ...More data for this Ligand-Target Pair
Affinity DataKi: 1.27E+4nMAssay Description:NK1: The affinity of compounds of the invention for the NK1 receptor was evaluated in CHO recombinant cells which express the human NK1 receptor. Mem...More data for this Ligand-Target Pair
Affinity DataKi: 1.40E+4nMAssay Description:NK2: Binding assays were performed in a 25 mM HEPES/1 mM CaCl2/5 mM MgCl2/0.5% BSA/10 μg/ml saponin, at pH 7.4. Binding assays consisted of 25 &...More data for this Ligand-Target Pair
Affinity DataKi: 1.40E+4nM ΔG°: -6.62kcal/molepH: 7.4 T: 2°CAssay Description:The affinity of compounds of the invention for the NK2 receptor was evaluated in CHO recombinant cells which express the human NK2 receptor. Membrane...More data for this Ligand-Target Pair
Affinity DataKi: 1.40E+4nMAssay Description:NK2: The affinity of compounds of the invention for the NK2 receptor was evaluated in CHO recombinant cells which express the human NK2 receptor. Mem...More data for this Ligand-Target Pair
Affinity DataKi: 1.40E+4nM ΔG°: -6.62kcal/molepH: 7.4 T: 2°CAssay Description:The affinity of compounds of the invention for the NK2 receptor was evaluated in CHO recombinant cells which express the human NK2 receptor. Membrane...More data for this Ligand-Target Pair
Affinity DataIC50: >3.00E+4nMAssay Description:hERG: The hERG inhibition study aims at quantifying the in vitro effects of compounds of the invention on the potassium-selective IKr current generat...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibition of CYP3A4 (unknown origin) by luciferase reporter assay in presence of NADPH regeneration systemMore data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:Antagonist activity at recombinant human NK3R expressed in CHO cells assessed as inhibition of NKB-induced Ca2+ signaling by aequorin Ca2+ biolumines...More data for this Ligand-Target Pair
Affinity DataIC50: >3.00E+4nMAssay Description:The hERG inhibition study aims at quantifying the in vitro effects of compounds of the invention on the potassium-selective IKf current generated in ...More data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:The antagonist activity of compounds of the invention is measured following pre-incubation (3 minutes) of the compound with the cells, followed by ad...More data for this Ligand-Target Pair
Affinity DataIC50: >3.00E+4nMAssay Description:The human Ether-a-go-go Related Gene (hERG) encodes the inward rectifying voltage gated potassium channel in the heart (IKr) which is involved in car...More data for this Ligand-Target Pair
Affinity DataIC50: 3.40E+4nMAssay Description:Inhibition of CYP2C19 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 2.40E+4nMAssay Description:Inhibition of human ERGMore data for this Ligand-Target Pair
Affinity DataIC50: 2.60E+4nMAssay Description:Inhibition of CYP1A2 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 2.10E+4nMAssay Description:Inhibition of CYP2C9 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of CYP2D6 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:Changes in intracellular calcium levels are a recognized indicator of G protein-coupled receptor activity. The efficacy of compounds of the invention...More data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:Antagonist activity against human recombinant NK3R expressed in CHO cells by aequorin functional assayMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibition of CYP3A4 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 2.40E+4nMAssay Description:Inhibition of human ERG expressed in HEK293 cells after 5 mins by patch-clamp assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2.60E+4nMAssay Description:Inhibition of CYP1A2 (unknown origin) by luciferase reporter assay in presence of NADPH regeneration systemMore data for this Ligand-Target Pair
Affinity DataIC50: 3.40E+4nMAssay Description:Inhibition of CYP2C19 (unknown origin) by luciferase reporter assay in presence of NADPH regeneration systemMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of CYP2D6 (unknown origin) by luciferase reporter assay in presence of NADPH regeneration systemMore data for this Ligand-Target Pair
Affinity DataIC50: 2.10E+4nMAssay Description:Inhibition of CYP2C9 (unknown origin) by luciferase reporter assay in presence of NADPH regeneration systemMore data for this Ligand-Target Pair