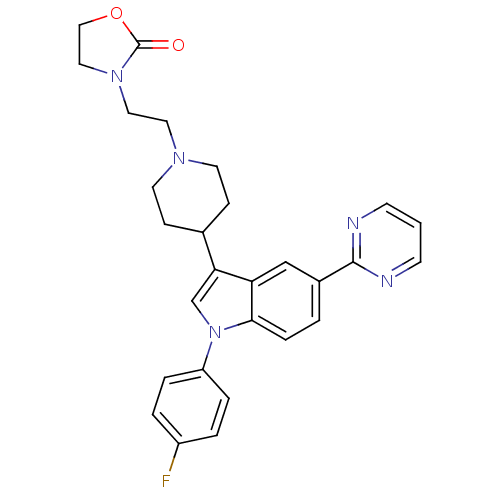
BDBM50122808 3-(2-{4-[1-(4-Fluoro-phenyl)-5-pyrimidin-2-yl-1H-indol-3-yl]-piperidin-1-yl}-ethyl)-oxazolidin-2-one::CHEMBL95175
SMILES Fc1ccc(cc1)-n1cc(C2CCN(CCN3CCOC3=O)CC2)c2cc(ccc12)-c1ncccn1
InChI Key InChIKey=VROFQJNNYWWTGM-UHFFFAOYSA-N
Data 11 KI
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 11 hits for monomerid = 50122808
Found 11 hits for monomerid = 50122808
Affinity DataKi: 0.560nMAssay Description:Displacement of [3H]prazosin (0.3 nM) from bovine Alpha-1A adrenergic receptor expressed in BHK cellsMore data for this Ligand-Target Pair
Affinity DataKi: 1.80nMAssay Description:Displacement of [3H]prazosin (0.5 nM) from hamster Alpha-1B adrenergic receptor expressed in rat-1 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 47nMAssay Description:Ability to displace [3H]5-CT (2.0 nM) from HeLa cells of human 5-hydroxytryptamine 1A receptorMore data for this Ligand-Target Pair
Affinity DataKi: 52nMAssay Description:Displacement of [3H]prazosin (0.3 nM) from rat Alpha-1D adrenergic receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 260nMAssay Description:Displacement of [3H]ketanserin (0.5 nM) from rat cerebral cortex 5-hydroxytryptamine 2A receptorsMore data for this Ligand-Target Pair
Affinity DataKi: 360nMAssay Description:Ability to displace [3H]5-CT (1.5 nM) from HeLa cells of human 5-hydroxytryptamine 1B receptorMore data for this Ligand-Target Pair
Affinity DataKi: 400nMAssay Description:Displacement of [3H]spiperone (0.5 nM) from rat corpus striatum dopamine D2 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 1.80E+3nMAssay Description:Ability to displace [3H]-SCH- 23390 (0.2 nM) from corpus striatum of rat Dopamine receptor D1More data for this Ligand-Target Pair
Affinity DataKi: 2.80E+3nMAssay Description:Ability to displace [3H]spiperone (0.3 nM) from CHO cells of human Dopamine receptor D3More data for this Ligand-Target Pair
Affinity DataKi: >4.20E+3nMAssay Description:Ability to displace [3H]mesulergine (0.5 nM) from CHO cells of human 5-hydroxytryptamine 2C receptorMore data for this Ligand-Target Pair
Affinity DataKi: 7.50E+3nMAssay Description:Displacement of [3H]-YM-09151-2 (0.06 nM) from human Dopamine receptor D4 expressed in CHO cellsMore data for this Ligand-Target Pair