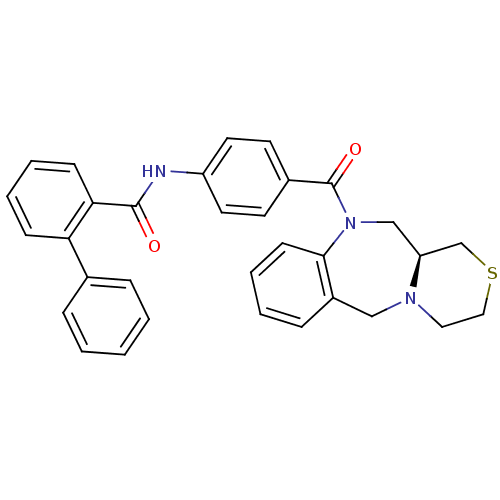
BDBM50146285 Biphenyl-2-carboxylic acid [4-((S)-3,4,11,11a-tetrahydro-1H,5H-2-thia-4a,10-diaza-dibenzo[a,d]cycloheptene-10-carbonyl)-phenyl]-amide::CHEMBL329903
SMILES O=C(Nc1ccc(cc1)C(=O)N1C[C@H]2CSCCN2Cc2ccccc12)c1ccccc1-c1ccccc1
InChI Key InChIKey=BXOKIJAEZHPQKJ-MHZLTWQESA-N
Data 4 KI
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50146285
Found 4 hits for monomerid = 50146285
TargetVasopressin V2 receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 3.20nMAssay Description:Inhibition of [3H]AVP binding to recombinant human vasopressin V2 receptorMore data for this Ligand-Target Pair
TargetVasopressin V2 receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 15nMAssay Description:Inhibition of 1 nM AVP-induced cAMP accumulation in cells expressing human vasopressin V2 receptorMore data for this Ligand-Target Pair
TargetVasopressin V1a receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 84nMAssay Description:Inhibition of [3H]AVP binding to recombinant human vasopressin V1a receptorMore data for this Ligand-Target Pair
TargetVasopressin V1a receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 1.40E+3nMAssay Description:Inhibition of 1 nM AVP-induced calcium mobilisation in cells expressing human vasopressin V1a receptorMore data for this Ligand-Target Pair