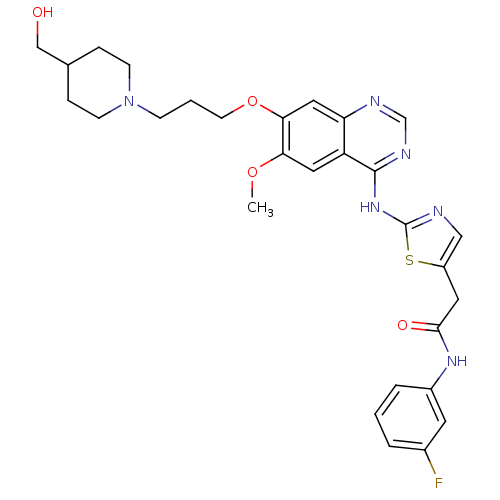
BDBM50181532 CHEMBL201307::N-(3-fluorophenyl)-2-(2-(7-(3-(4-(hydroxymethyl)piperidin-1-yl)propoxy)-6-methoxyquinazolin-4-ylamino)thiazol-5-yl)acetamide::N-(3-fluorophenyl)-2-{2-[(7-{3-[4-(hydroxymethyl)piperidin-1-yl]propoxy}-6-methoxyquinazolin-4-yl)amino]-1,3-thiazol-5-yl}acetamide::N-3-fluorophenyl)-2-2-7-3-4-hydroxymethyl)piperidin-1-yl)propoxy)-6-methoxyquinazolin-4-ylamino)thiazol-5-yl)acetamide
SMILES COc1cc2c(Nc3ncc(CC(=O)Nc4cccc(F)c4)s3)ncnc2cc1OCCCN1CCC(CO)CC1
InChI Key InChIKey=QNJAKUMXJFWQOG-UHFFFAOYSA-N
Data 18 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 18 hits for monomerid = 50181532
Found 18 hits for monomerid = 50181532
Affinity DataIC50: 7nMAssay Description:Inhibition of Aurora B kinaseMore data for this Ligand-Target Pair
Affinity DataIC50: 7nMAssay Description:Inhibition of aurora B kinaseChecked by AuthorMore data for this Ligand-Target Pair
Affinity DataIC50: <1nMAssay Description:Inhibition of Aurora A kinase activityMore data for this Ligand-Target Pair
TargetcAMP-dependent protein kinase catalytic subunit alpha/beta/gamma(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory activity against PKAMore data for this Ligand-Target Pair
Affinity DataIC50: 8.00E+3nMAssay Description:Inhibitory activity against FGFRMore data for this Ligand-Target Pair
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Novartis Research Foundation
Curated by ChEMBL
Novartis Research Foundation
Curated by ChEMBL
Affinity DataIC50: 800nMAssay Description:Inhibition of EGFR kinaseMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory activity against p38-alpha MAPKMore data for this Ligand-Target Pair
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Novartis Research Foundation
Curated by ChEMBL
Novartis Research Foundation
Curated by ChEMBL
Affinity DataIC50: 800nMAssay Description:Inhibitory activity against EGFRMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibitory activity against SRCMore data for this Ligand-Target Pair
Affinity DataIC50: 2.50E+4nMAssay Description:Inhibitory activity against JAK3More data for this Ligand-Target Pair
Affinity DataIC50: 4.00E+3nMAssay Description:Inhibitory activity against FAKMore data for this Ligand-Target Pair
Affinity DataIC50: 2.10E+4nMAssay Description:Inhibitory activity against CSKMore data for this Ligand-Target Pair
TargetVascular endothelial growth factor receptor 2(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibitory activity against KDRMore data for this Ligand-Target Pair
Affinity DataIC50: 1.30E+4nMAssay Description:Inhibitory activity against IGFRMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory activity against JNK1alphaMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibitory activity against human hERG receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 6.20E+4nMAssay Description:Inhibitory activity against CDK2More data for this Ligand-Target Pair
Affinity DataIC50: <1nMAssay Description:Inhibition of Aurora A kinaseMore data for this Ligand-Target Pair