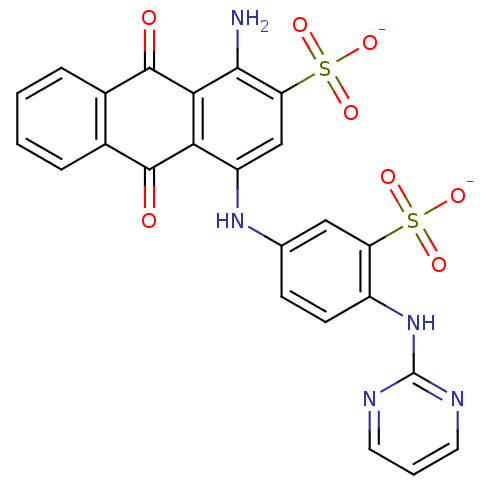
BDBM50268965 CHEMBL499428::Disodium 1-amino-4-[4-([1,3]diazine-2-ylamino)-3-sulfophenylamino]-9,10-dioxo-9,10-dihydroanthracene-2-sulfonate
SMILES Nc1c(cc(Nc2ccc(Nc3ncccn3)c(c2)S([O-])(=O)=O)c2C(=O)c3ccccc3C(=O)c12)S([O-])(=O)=O
InChI Key InChIKey=ZIFQNSIXEIWWPD-UHFFFAOYSA-L
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50268965
Found 4 hits for monomerid = 50268965
Affinity DataKi: 50.7nMAssay Description:Displacement of [3H]PSB0413 from human platelet P2Y12 receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:Antagonist activity against rat P2X2 receptor expressed in Xenopus laevis oocyte assessed as inhibition of alpha, beta-meATP-induced inward current b...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Antagonist activity against rat P2X4 receptor expressed in Xenopus laevis oocyte assessed as inhibition of alpha, beta-meATP-induced inward current b...More data for this Ligand-Target Pair
Affinity DataIC50: 760nMAssay Description:Displacement of [3H]DPCPX from human adenosine A1 receptor expressed in CHO cells assessed as ratio of IC50 in presence of GTP to absence of GTP by G...More data for this Ligand-Target Pair