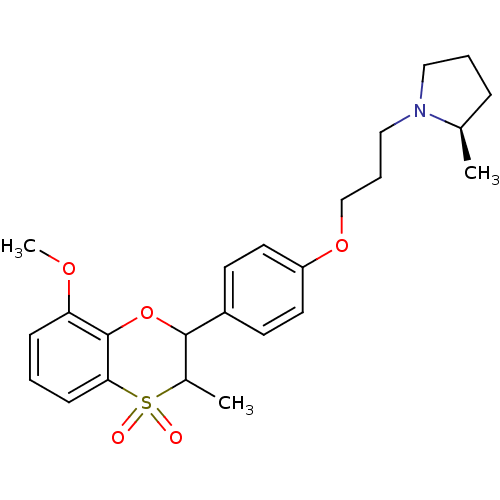
BDBM50296176 (+/-)-(R)-1-{3-[4-(8-Methoxy-3-methyl-4,4-dioxo-3,4-dihydro-2H-4lambda*6*-benzo[1,4]oxathiin-2-yl)-phenoxy]-propyl}-2-methyl-pyrrolidine::CHEMBL560863
SMILES COc1cccc2c1OC(C(C)S2(=O)=O)c1ccc(OCCCN2CCC[C@H]2C)cc1
InChI Key InChIKey=FWYZZPBECMKUCM-GBFLJEJBSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50296176
Found 4 hits for monomerid = 50296176
Affinity DataKi: 0.200nMAssay Description:Displacement of [125I]Iodoproxyfan from human recombinant histamine H3 receptor by Competitive binding assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Bioprojet-Biotech
Curated by ChEMBL
Bioprojet-Biotech
Curated by ChEMBL
Affinity DataIC50: 2.90E+3nMAssay Description:Displacement of [3H]dofetilide from human recombinant ERG by Competitive binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 0.210nMAssay Description:Inverse agonist activity at human cloned histamine H3 receptor assessed as inhibition of R-alpha-methylistamine-induced [35S]GTPgammaS binding by cel...More data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Bioprojet-Biotech
Curated by ChEMBL
Bioprojet-Biotech
Curated by ChEMBL
Affinity DataIC50: 2.90E+3nMAssay Description:Inhibition of [35S]MK499 binding to human ERG transfected in HEK293 cells by microscintillation countingMore data for this Ligand-Target Pair