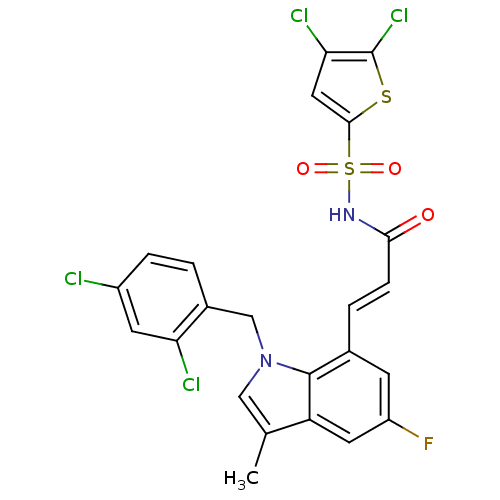
BDBM50303662 3-(1-(2,4-dichlorobenzyl)-5-fluoro-3-methyl-1H-indol-7-yl)-N-(4,5-dichlorothiophen-2-ylsulfonyl)acrylamide::CHEMBL565591
SMILES Cc1cn(Cc2ccc(Cl)cc2Cl)c2c(\C=C\C(=O)NS(=O)(=O)c3cc(Cl)c(Cl)s3)cc(F)cc12
InChI Key InChIKey=BFBTVZNKWXWKNZ-HWKANZROSA-N
Data 15 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 15 hits for monomerid = 50303662
Found 15 hits for monomerid = 50303662
Affinity DataIC50: 1.50E+3nMAssay Description:Inhibition of human CYP3A4 using 7-benzyloxy-4-trifluoromethylcoumarin as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 4.60nMAssay Description:Displacement of [3H]PGE2 from human EP3 receptorMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of human CYP2D6More data for this Ligand-Target Pair
Affinity DataIC50: 30nMAssay Description:Inhibition of human CYP2C9More data for this Ligand-Target Pair
Affinity DataIC50: 6.60E+3nMAssay Description:Inhibition of human CYP2C19More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of human CYP1A2More data for this Ligand-Target Pair
Affinity DataIC50: 11nMAssay Description:Displacement of [3H]PGE2 from mouse EP3 receptor after 1 hr by liquid scintillation countingMore data for this Ligand-Target Pair
Affinity DataIC50: 1.44E+4nMAssay Description:Displacement of [3H]iloprost from human IP receptor after 1 hr by liquid scintillation countingMore data for this Ligand-Target Pair
Affinity DataIC50: 8.04E+3nMAssay Description:Displacement of [3H]PGE2 from human EP4 receptor after 1 hr by liquid scintillation countingMore data for this Ligand-Target Pair
Affinity DataIC50: 4.17E+3nMAssay Description:Displacement of [3H]PGE2 from human EP2 receptor after 1 hr by liquid scintillation countingMore data for this Ligand-Target Pair
Affinity DataIC50: >2.00E+4nMAssay Description:Displacement of [3H]PGE2 from human EP1 receptor after 1 hr by liquid scintillation countingMore data for this Ligand-Target Pair
Affinity DataIC50: 4.60nMAssay Description:Displacement of [3H]PGE2 from human EP3 receptor after 60 mins repeated washing by liquid scintillation countingMore data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Displacement of [3H]PGE2 from human EP3 receptor after 1 hr by liquid scintillation counting in presence of 10% human serumMore data for this Ligand-Target Pair
Affinity DataIC50: 4.60nMAssay Description:Displacement of [3H]PGE2 from human EP3 receptor after 1 hr by liquid scintillation countingMore data for this Ligand-Target Pair
Affinity DataIC50: 7.60E+3nMAssay Description:Inhibition of human CYP3A4 using dibenzylfluorescein as substrateMore data for this Ligand-Target Pair