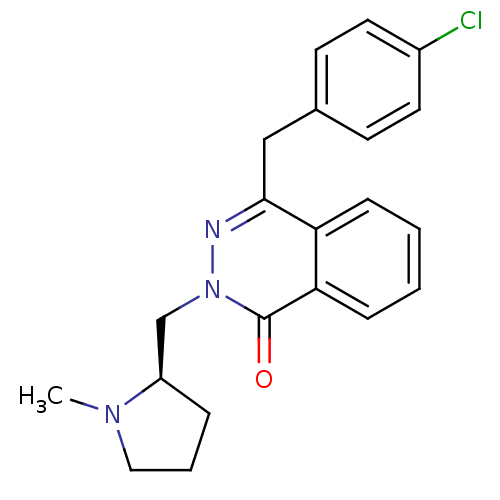
BDBM50391708 CHEMBL1767136
SMILES CN1CCC[C@@H]1Cn1nc(Cc2ccc(Cl)cc2)c2ccccc2c1=O
InChI Key InChIKey=WYRUGBCTQBZIHO-QGZVFWFLSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 50391708
Found 6 hits for monomerid = 50391708
Affinity DataKi: 0.158nMAssay Description:Antagonist activity at human recombinant histamine H1 receptor expressed in intact CHO cells assessed as inhibition of histamine-induced cellular cal...More data for this Ligand-Target Pair
Affinity DataKi: 0.162nMAssay Description:Antagonist activity at human H1 receptor expressed in CHO cells assessed as inhibition of calcium mobilization by FLIPR assayMore data for this Ligand-Target Pair
Affinity DataKi: 10nMAssay Description:Antagonist activity at human adrenergic alpha1B receptor expressed in rat fibroblasts by by plate-based calcium imagingMore data for this Ligand-Target Pair
Affinity DataKi: 31.6nMAssay Description:Antagonist activity at human adrenergic alpha1A receptor expressed in rat fibroblasts by by plate-based calcium imagingMore data for this Ligand-Target Pair
Affinity DataKi: 437nMAssay Description:Antagonist activity at human H3 receptor expressed in CHO cells assessed as inhibition of histamine-induced GTPgamma[S] binding by scintillation prox...More data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Glaxosmithkline
Curated by ChEMBL
Glaxosmithkline
Curated by ChEMBL
Affinity DataIC50: 200nMAssay Description:Displacement of [3H]dofetilide from human ERGMore data for this Ligand-Target Pair