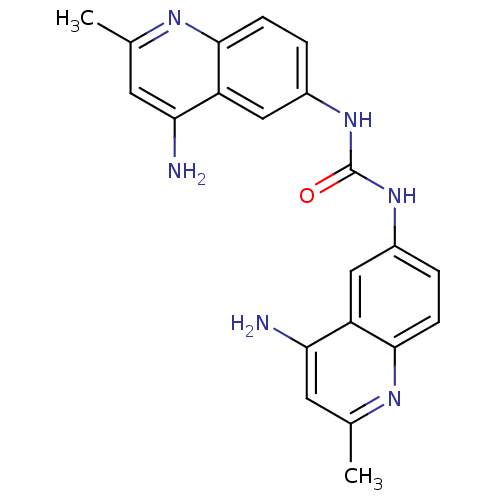
BDBM50444748 Aminoquinuride
SMILES Cc1cc(N)c2cc(NC(=O)Nc3ccc4nc(C)cc(N)c4c3)ccc2n1
InChI Key InChIKey=HOUSDILKOJMENG-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 50444748
Found 6 hits for monomerid = 50444748
Affinity DataKi: 500nMAssay Description:Competitive inhibition of Bacillus anthracis lethal factorMore data for this Ligand-Target Pair
Affinity DataIC50: 3.20E+3nMAssay Description:Inhibition of Bacillus anthracis lethal factor assessed as MCA-KKVYPYPME[dnp]K amide cleavage after 30 mins by fluorescence plate reader analysisMore data for this Ligand-Target Pair
TargetGuanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2/G(I)/G(S)/G(T) subunit beta-1(Homo sapiens)
Sun Yat-Sen University
Curated by ChEMBL
Sun Yat-Sen University
Curated by ChEMBL
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibition of G-protein beta1gamma2 (unknown origin) by flow cytometryMore data for this Ligand-Target Pair
TargetC5a anaphylatoxin chemotactic receptor 1(Homo sapiens (Human))
Merck Sharp And Dohme Research Laboratories
Curated by ChEMBL
Merck Sharp And Dohme Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 8.86E+3nMAssay Description:Ability to inhibit binding of radiolabeled C5a anaphylatoxin to intact membrane bound receptor from human neutrophils.More data for this Ligand-Target Pair
TargetMyeloperoxidase(Homo sapiens (Human))
Merck Sharp And Dohme Research Laboratories
Curated by ChEMBL
Merck Sharp And Dohme Research Laboratories
Curated by ChEMBL
Affinity DataEC50: <3.00E+3nMAssay Description:Compound was tested for the induction of nonspecific myeloperoxidase (MPO) releaseMore data for this Ligand-Target Pair
Affinity DataIC50: 7.20E+3nMAssay Description:Inhibition of Clostridium bolulinum BoNT/A assessed as cleavage of MOCAc-Lys-Lys-Val-Tyr-Pro-Tyr-Pro-Met-Glu-Lys(Dnp)-NH2 after 40 mins by FRET assayMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)