Affinity DataKi: 0.0130nM ΔG°: -61.5kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.0240nM ΔG°: -60.0kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.0280nM ΔG°: -59.6kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.0360nM ΔG°: -59.0kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.0440nM ΔG°: -58.5kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.0580nM ΔG°: -57.8kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.0800nM ΔG°: -57.1kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.0910nM ΔG°: -56.7kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.0940nM ΔG°: -56.7kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.100nM ΔG°: -56.5kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.120nM ΔG°: -56.1kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.190nM ΔG°: -54.9kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.200nM ΔG°: -54.8kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.25nM ΔG°: -54.3kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.260nM ΔG°: -54.2kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.270nM ΔG°: -54.1kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.270nM ΔG°: -54.1kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.290nM ΔG°: -53.9kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.310nM ΔG°: -53.7kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.380nM ΔG°: -53.2kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.420nM ΔG°: -53.0kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.580nM ΔG°: -52.2kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 0.650nM ΔG°: -51.9kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 4.20nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 5.10nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 5.40nM ΔG°: -46.7kJ/molepH: 7.0 T: 2°CAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 5.5nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 6.40nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 7.90nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 7.90nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 8nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 10nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 11nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 11nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 11nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 11nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 15nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 20nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 26nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 44nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 72nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 82nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 120nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 190nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 1.70E+3nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair
Affinity DataKi: 1.90E+3nMAssay Description:Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter...More data for this Ligand-Target Pair

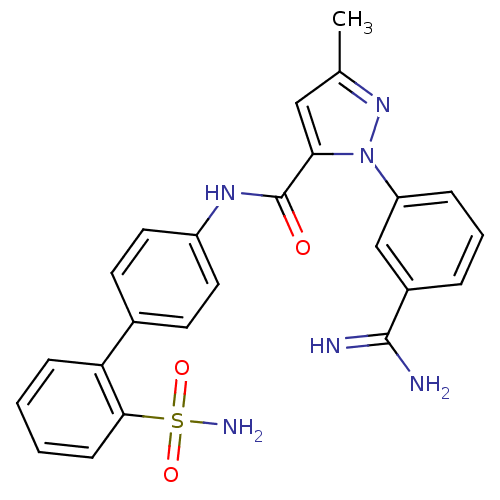
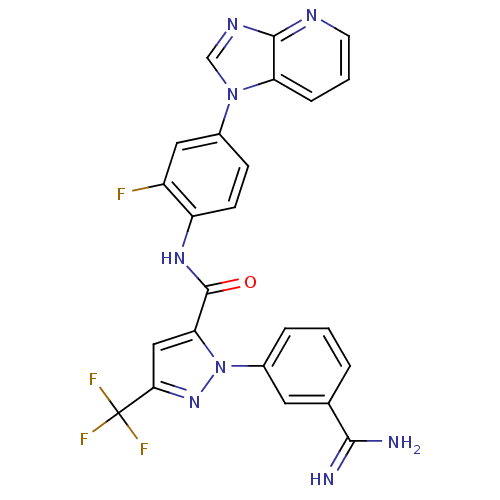
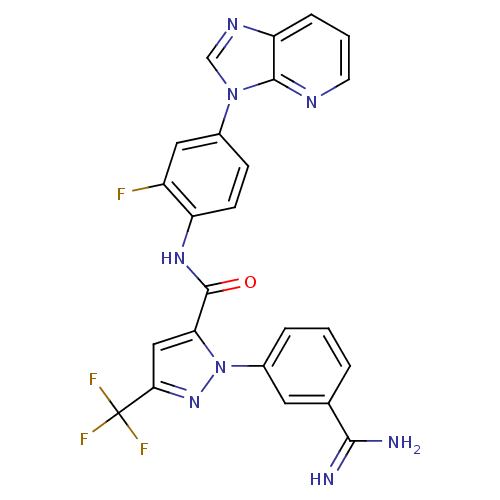
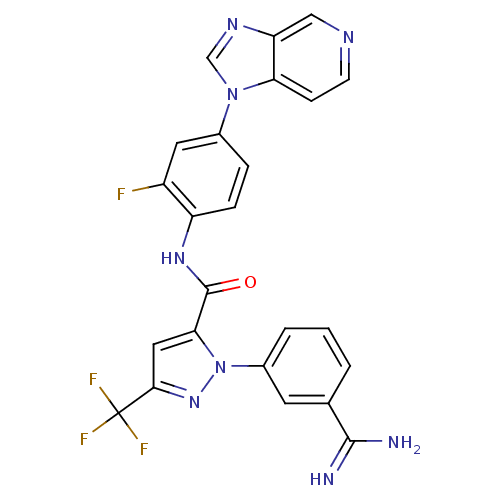
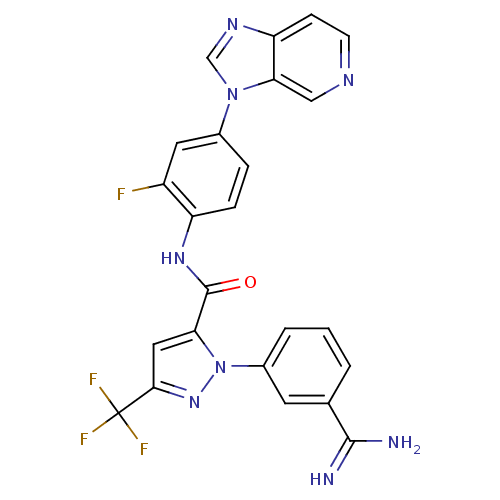
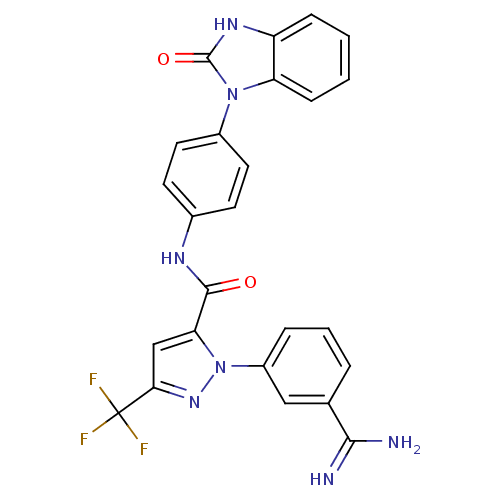
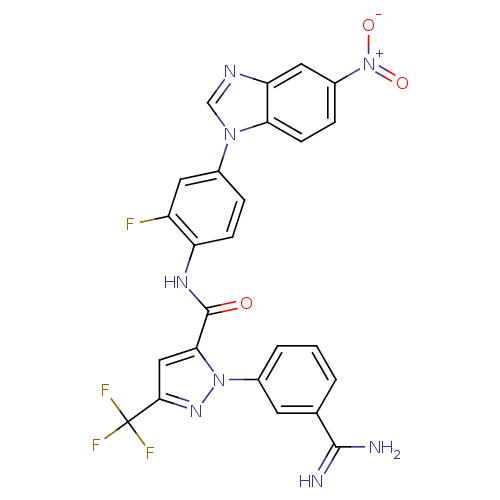
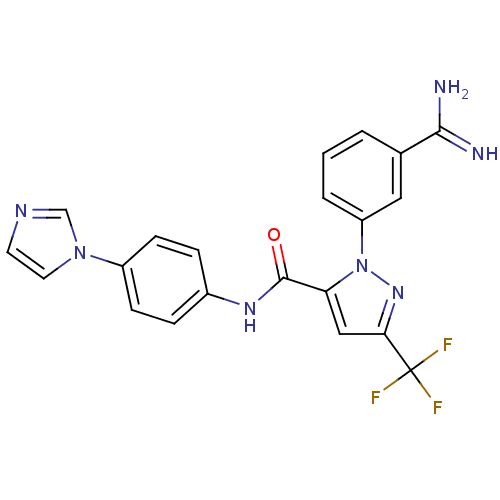
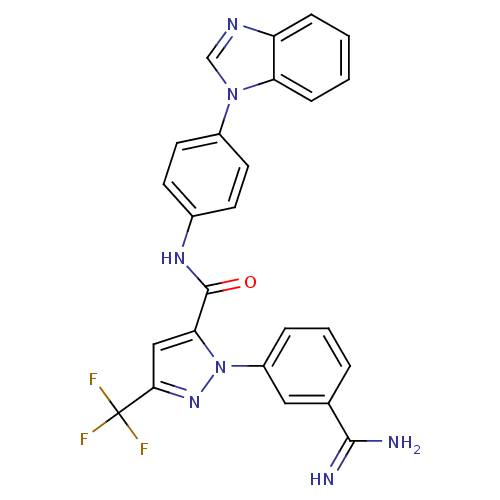
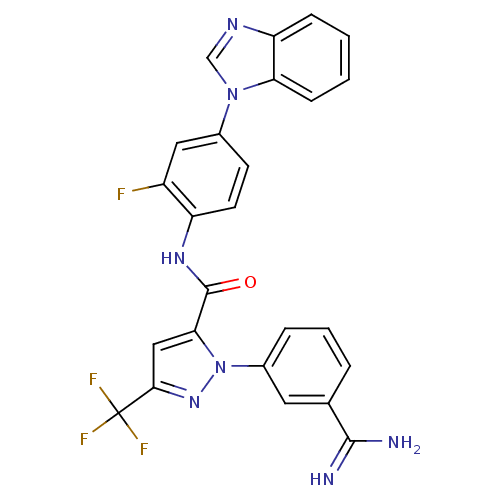
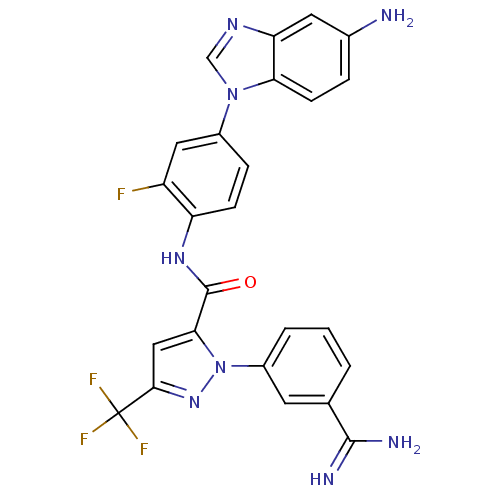
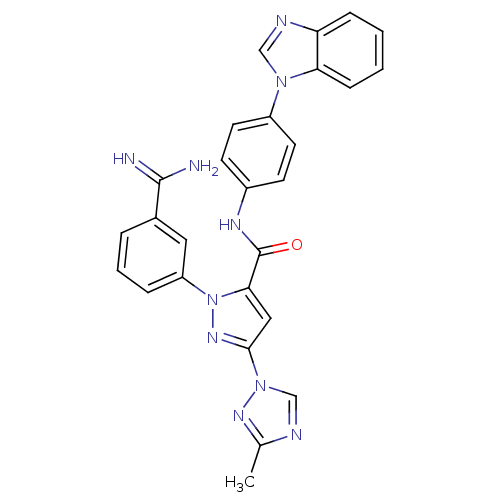
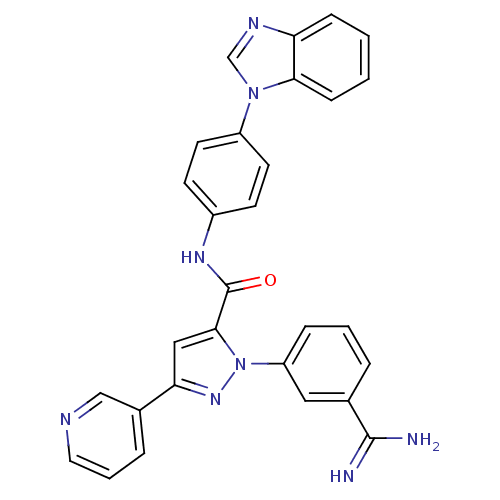
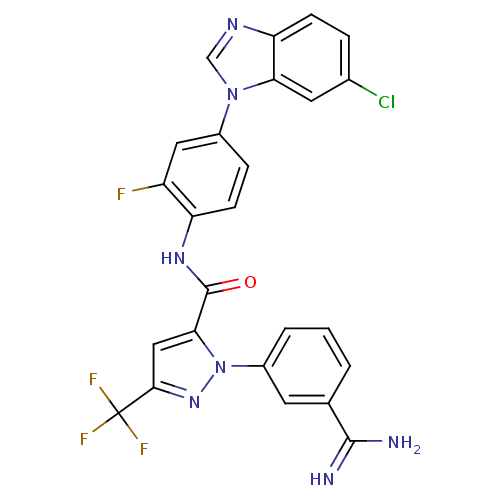
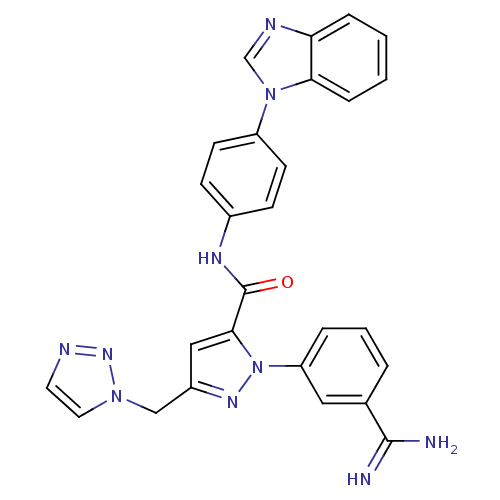
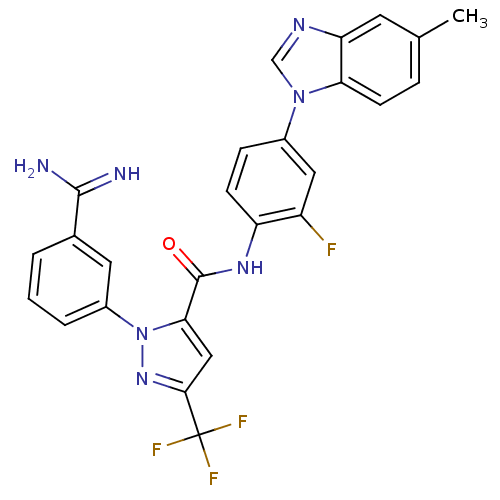

 3D Structure (crystal)
3D Structure (crystal)