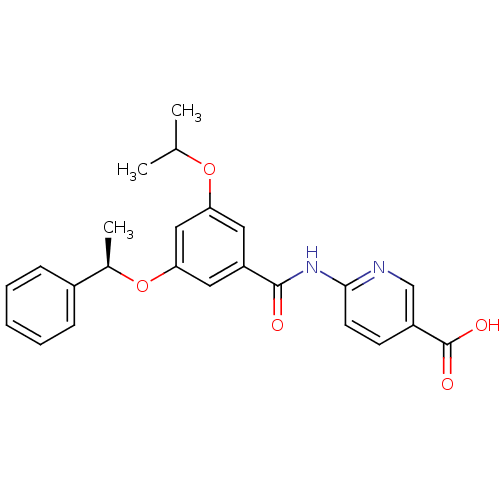
Affinity DataEC50: 5.51E+3nMAssay Description:Activitation of glucokinaseMore data for this Ligand-Target Pair
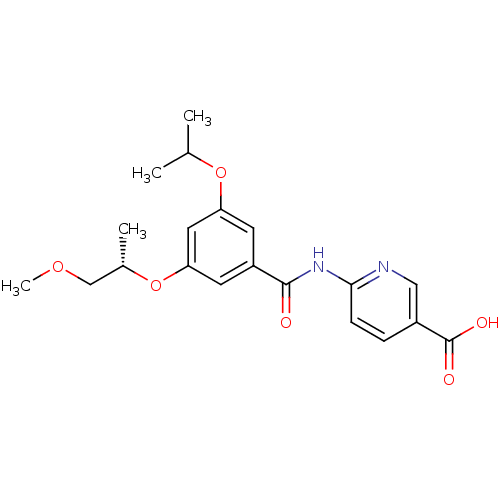
Affinity DataEC50: 950nMAssay Description:Activitation of glucokinaseMore data for this Ligand-Target Pair
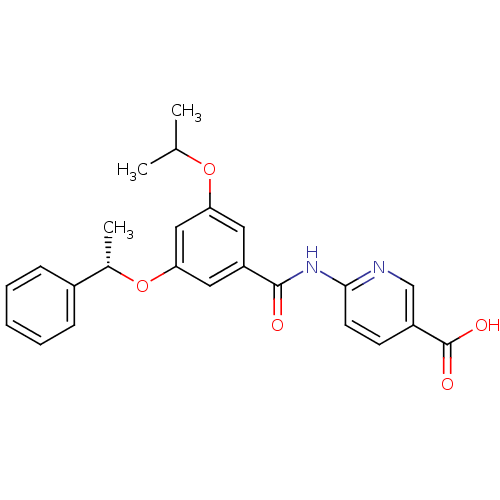
Affinity DataEC50: 610nMAssay Description:Activitation of glucokinaseMore data for this Ligand-Target Pair
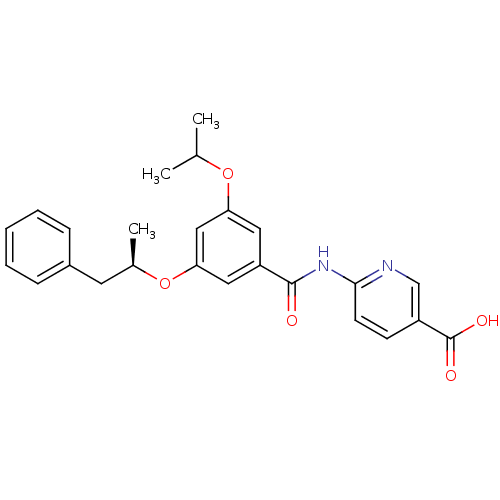
Affinity DataEC50: 20nMAssay Description:Activitation of glucokinaseMore data for this Ligand-Target Pair
Affinity DataEC50: 110nMAssay Description:Activitation of glucokinaseMore data for this Ligand-Target Pair
Affinity DataEC50: 90nMAssay Description:Activitation of glucokinaseMore data for this Ligand-Target Pair