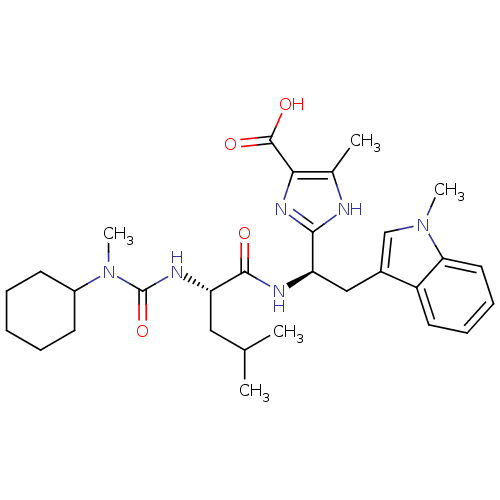
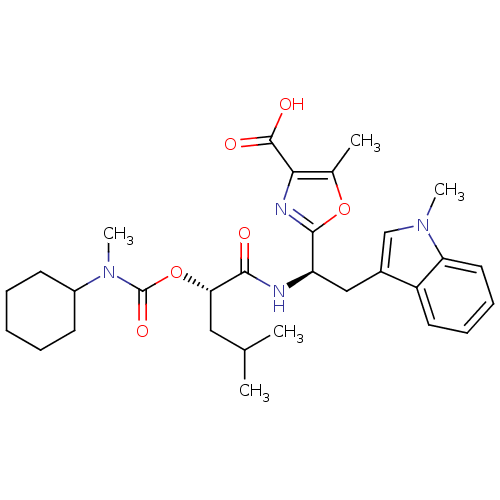
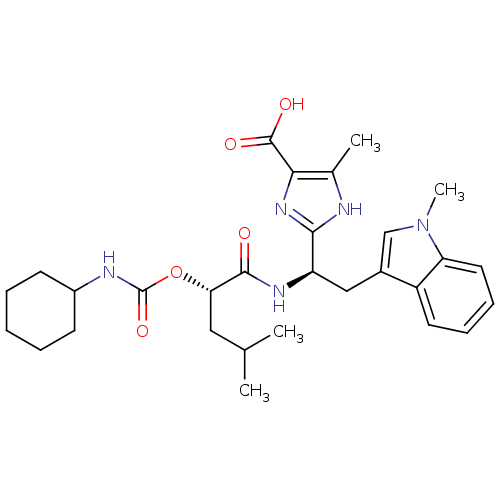
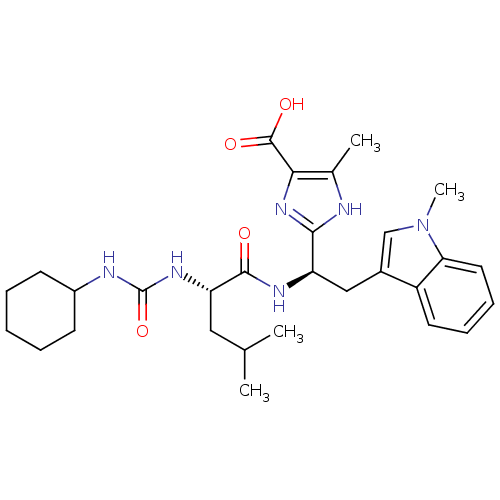
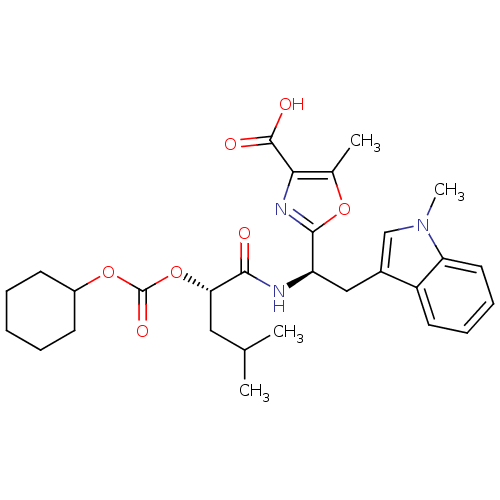
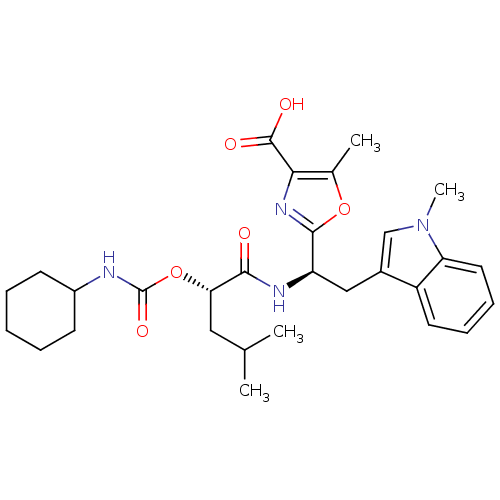
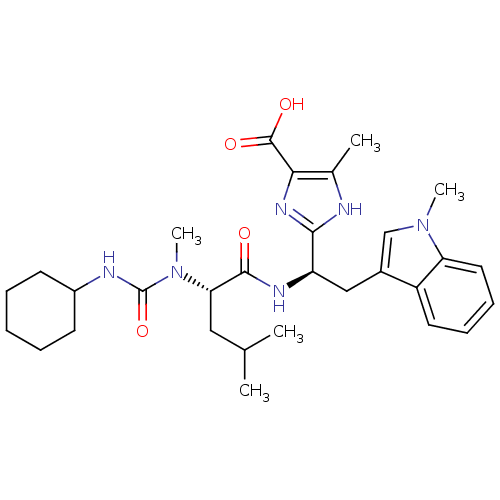
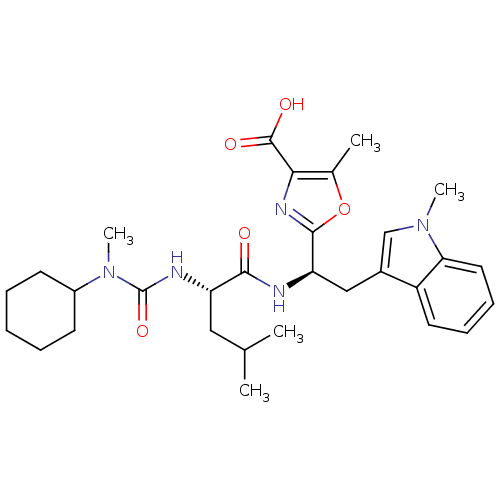
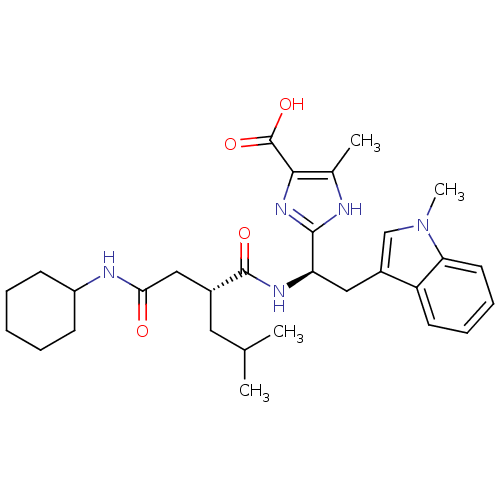
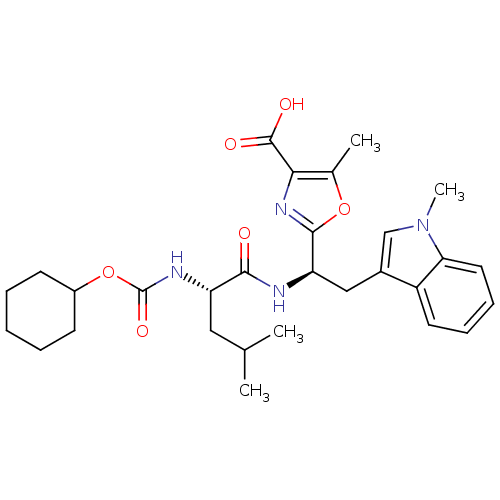
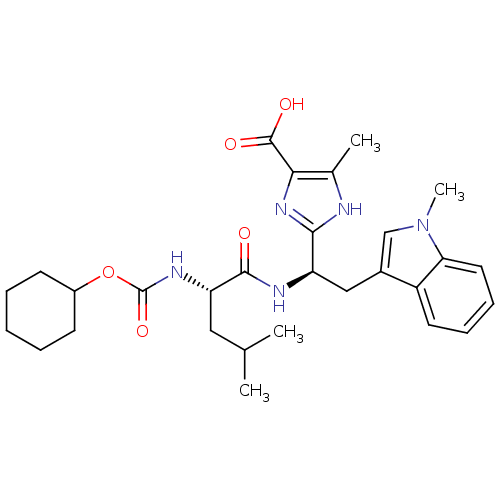
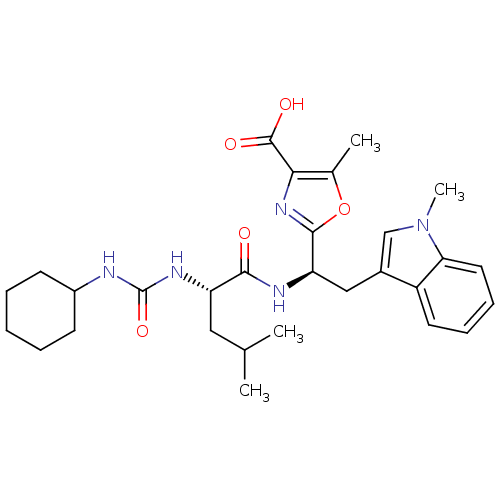
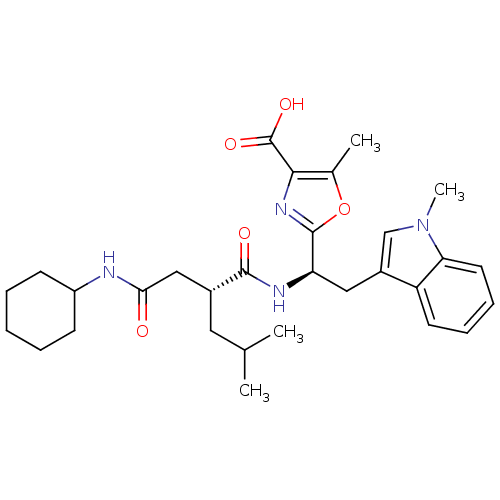
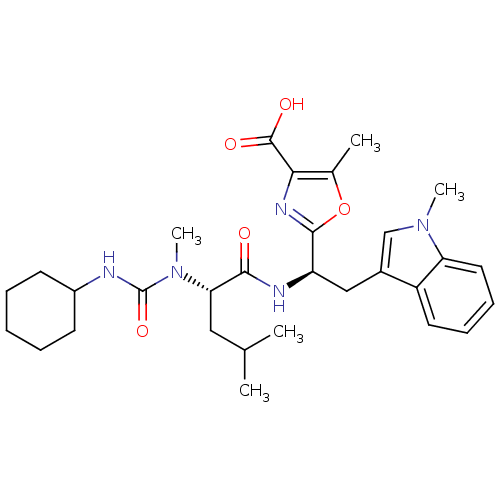
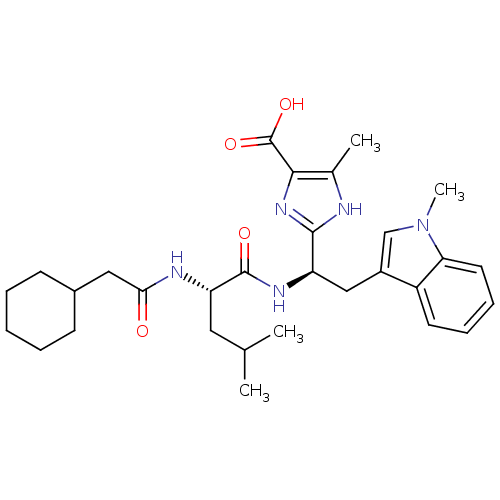
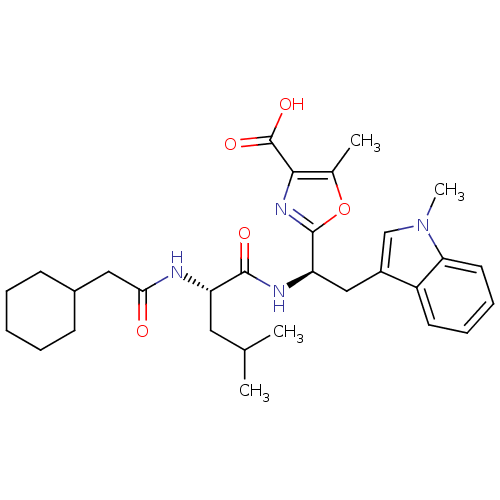
Affinity DataIC50: 1.80nMAssay Description:Inhibitory activity was evaluated against human Endothelin A receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 4.60nMAssay Description:Inhibitory activity was evaluated against human Endothelin A receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 9.30nMAssay Description:Inhibitory activity was evaluated against human Endothelin A receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 11nMAssay Description:Inhibitory activity was evaluated against human Endothelin A receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 17nMAssay Description:Inhibitory activity was evaluated against human Endothelin A receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 21nMAssay Description:Inhibitory activity was evaluated against human Endothelin A receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 22nMAssay Description:Inhibitory activity was evaluated against human Endothelin A receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 25nMAssay Description:Inhibitory activity was evaluated against human Endothelin A receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 29nMAssay Description:Inhibitory activity was evaluated against human Endothelin A receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 41nMAssay Description:Inhibitory activity was evaluated against human Endothelin A receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 76nMAssay Description:Inhibitory activity was evaluated against human Endothelin A receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 220nMAssay Description:Inhibitory activity was evaluated against human Endothelin A receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 310nMAssay Description:Inhibitory activity was evaluated against human Endothelin A receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 450nMAssay Description:Inhibitory activity was evaluated against human Endothelin A receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 470nMAssay Description:Inhibitory activity was evaluated against human Endothelin A receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 680nMAssay Description:Inhibitory activity was evaluated against human Endothelin A receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+4nMAssay Description:Inhibitory activity was evaluated against human Endothelin B receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 3.40E+4nMAssay Description:Inhibitory activity was evaluated against human Endothelin B receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 4.80E+4nMAssay Description:Inhibitory activity was evaluated against human Endothelin B receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 5.40E+4nMAssay Description:Inhibitory activity was evaluated against human Endothelin B receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 7.90E+4nMAssay Description:Inhibitory activity was evaluated against human Endothelin B receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 9.00E+4nMAssay Description:Inhibitory activity was evaluated against human Endothelin B receptorMore data for this Ligand-Target Pair
Affinity DataIC50: >9.00E+4nMAssay Description:Inhibitory activity was evaluated against human Endothelin B receptorMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory activity was evaluated against human Endothelin B receptorMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory activity was evaluated against human Endothelin B receptorMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory activity was evaluated against human Endothelin B receptorMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory activity was evaluated against human Endothelin B receptorMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory activity was evaluated against human Endothelin B receptorMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory activity was evaluated against human Endothelin B receptorMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory activity was evaluated against human Endothelin B receptorMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory activity was evaluated against human Endothelin B receptorMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory activity was evaluated against human Endothelin B receptorMore data for this Ligand-Target Pair