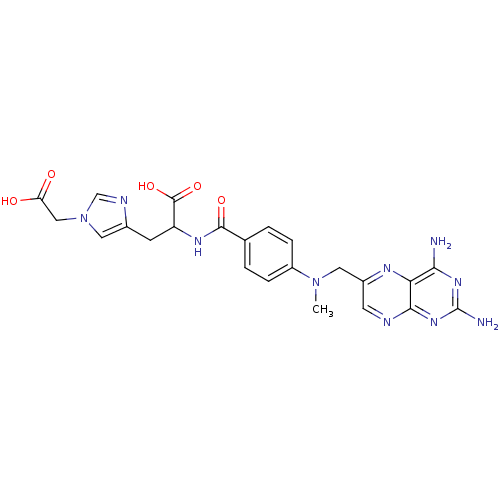
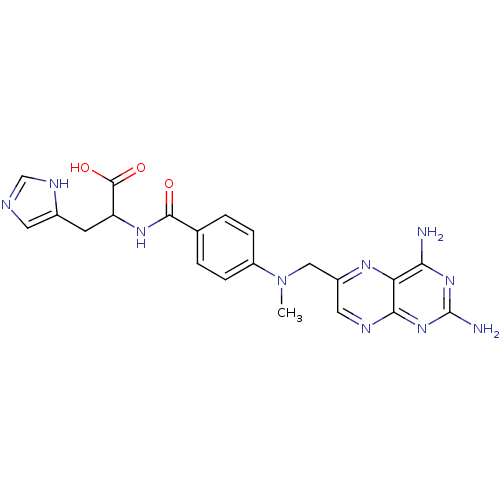
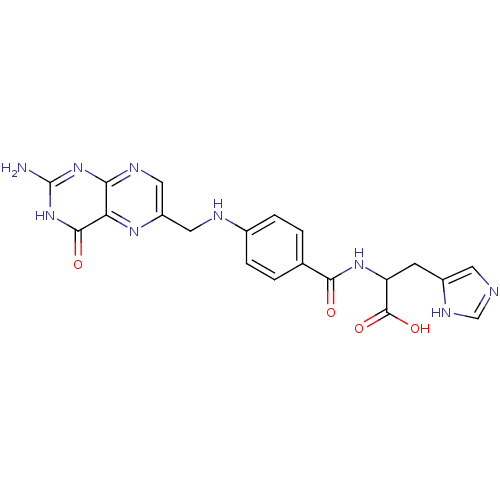
Affinity DataIC50: 73nMAssay Description:compound was evaluated for the inhibitory activity against Dihydrofolate reductase in permeabilised L1210 cells.More data for this Ligand-Target Pair
Affinity DataIC50: 370nMAssay Description:compound was evaluated for the inhibitory activity against Dihydrofolate reductase in permeabilised L1210 cells.More data for this Ligand-Target Pair
Affinity DataIC50: 1.30E+3nMAssay Description:compound was evaluated for the inhibitory activity against Dihydrofolate reductase in permeabilised L1210 cells.More data for this Ligand-Target Pair
TargetFolylpolyglutamate synthase, mitochondrial(Homo sapiens (Human))
State University Of New York
Curated by ChEMBL
State University Of New York
Curated by ChEMBL
Affinity DataIC50: 3.20E+3nMAssay Description:Compound was evaluated for the inhibitory activity against Folyl-polyglutamate synthase from CCRF-CEM human leukemia cells.More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:compound was evaluated for the inhibitory activity against Dihydrofolate reductase in permeabilised L1210 cells.More data for this Ligand-Target Pair
TargetFolylpolyglutamate synthase, mitochondrial(Homo sapiens (Human))
State University Of New York
Curated by ChEMBL
State University Of New York
Curated by ChEMBL
Affinity DataIC50: >2.00E+5nMAssay Description:Compound was evaluated for the inhibitory activity against Folyl-polyglutamate synthase from CCRF-CEM human leukemia cells.More data for this Ligand-Target Pair
TargetFolylpolyglutamate synthase, mitochondrial(Homo sapiens (Human))
State University Of New York
Curated by ChEMBL
State University Of New York
Curated by ChEMBL
Affinity DataIC50: >2.00E+5nMAssay Description:Compound was evaluated for the inhibitory activity against Folyl-polyglutamate synthase from CCRF-CEM human leukemia cells.More data for this Ligand-Target Pair
TargetFolylpolyglutamate synthase, mitochondrial(Homo sapiens (Human))
State University Of New York
Curated by ChEMBL
State University Of New York
Curated by ChEMBL
Affinity DataIC50: >2.00E+5nMAssay Description:Compound was evaluated for the inhibitory activity against Folyl-polyglutamate synthase from CCRF-CEM human leukemia cells.More data for this Ligand-Target Pair

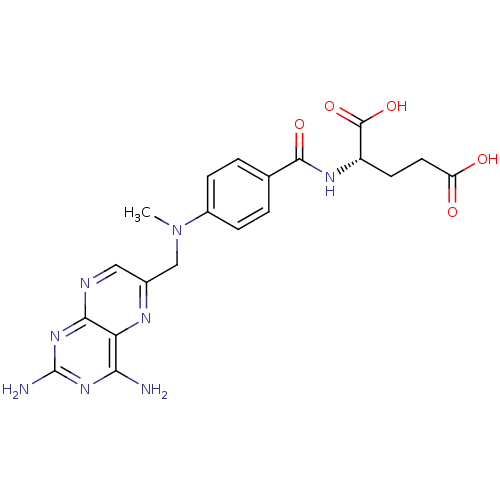
 3D Structure (crystal)
3D Structure (crystal)